Cheap 500 mg hydrea with amex
Smooth muscle in the uterus proliferates during the normal menstrual cycle and during pregnancy; both activities are under hormonal control symptoms melanoma safe 500mg hydrea. The smooth muscle cells of blood vessels also divide regularly in the adult, presumably to replace damaged or senile cells; the smooth muscle of the muscularis externa of the stomach and colon regularly replicates and may even slowly thicken during life. New smooth muscle cells have been shown to differentiate from undifferentiated mesenchymal stem cells in the adventitia of blood vessels. Differentiation of smooth muscle progenitor cells is regulated by a variety of intracellular and environmental stimuli, and developing muscles exhibit a wide range of different phenotypes at different stages of their development. To date, no transcription factors have been identified that are characteristic for the smooth muscle cell lineage. Smooth muscle cells have also been shown to develop from the division and differentiation of endothelial cells and pericytes during the repair process after vascular injury. Vascular pericytes are located within the basal lamina of capillaries and postcapillary venules. In capillaries, their cytoplasmic morphology is difficult to distinguish from that of the endothelial cell. In postcapillary venules and pericytic venules, they may form a nearly complete investment of the vessel with cells that resemble smooth muscle cells (see Chapter 13, Cardiovascular System). Fibroblasts in healing wounds may develop morphologic and functional characteristics of smooth muscle cells (myofibroblasts; see page 175). Epithelial cells in numerous locations, particularly sweat glands, mammary glands, salivary glands, and the iris of the eye, may acquire the characteristics of smooth muscle cells (myoepithelial cells). Myoid cells of the testis have a contractile function in the seminiferous tubules, and cells of the perineurium, a concentric layer of connective tissue that surrounds groups of nerve fibers and partitions peripheral nerves into distinct fascicles, function as contractile cells as well as transport barrier cells. These common characteristics suggest that cardiac muscle may have evolved in the direction of skeletal muscle from the smooth muscle of primitive circulatory systems. A summary of major characteristics of all three muscle types is provided in the table below. Cardiac muscle shares structural and functional characteristics with skeletal muscle and smooth muscle. Both cardiac and smooth muscle cells retain their individuality, although both are in functional communication with their neighbors through gap junctions. There are three major types of muscle tissue: skeletal, cardiac, and smooth muscle. Endomysium surrounds individual fibers; perimysium surrounds a group of fibers to form a fascicle; and epimysium is dense connective tissue that surrounds the entire muscle. Three types of skeletal muscle fibers are distinguished based on contractile speed, enzymatic velocity, and metabolic profile. It is composed of precisely aligned myofilaments: myosin-containing thick filaments and actin-containing thin filaments. The arrangement of thick and thin filaments gives rise to the density differences that produce the cross-striations of the myofibril. The light-staining isotropic I band contains mainly thin filaments attached to both sides of the Z line, and the dark-staining anisotropic A band contains mainly thick filaments. Z lines between sarcomeres contain an actin-binding protein (-actinin) and Z matrix proteins. The actomyosin cross-bridge cycle represents a series of coupled biochemical and mechanical events between myosin heads and actin molecules that lead to muscle contraction. There are five recognizable stages of the cycle: attachment, release, bending, force generation, and reattachment. Regulation of muscle contraction involves Ca2, sarcoplasmic reticulum, and the transverse tubular system. The sarcoplasmic reticulum forms enlarged terminal cisternae that serve as reservoirs for Ca2. Their plasma membrane contains an abundance of gated Ca2 -release channels (ryanodine receptors [RyR1]). The transverse tubules (T tubules) are formed by invaginations of the sarcoplasm that penetrate the muscle fiber between adjacent terminal cisternae. Triads are located at the junction between A and I bands (two per each sarcomere). The neuromuscular junction (motor end plate) is the contact area of the axon endings with muscle fiber. Encapsulated muscle spindles and Golgi tendon organs are sensory (proprioreceptive) stretch receptors in muscles and tendons. Cardiac muscle cells (cardiac myocytes) are short cylindrical cells with a centrally positioned single nucleus. They are attached to each other by intercalated discs to form a cardiac muscle fiber. The intercalated discs represent highly specialized cell-to-cell adhesion junctions containing fascia adherens, gap junctions, and maculae adherentes (desmosomes). Terminal cisternae are much smaller than in skeletal muscle and with the T tubules form diads that are located at the level of Muscle Tissue the Z line (one per sarcomere). Passage of Ca2 from the lumen of the T tubule to the sarcoplasm of a cardiac myocyte is essential to initiate the contraction cycle. Specialized cardiac conducting muscle cells (Purkinje fibers) exhibit a spontaneous rhythmic contraction. They generate and rapidly transmit action potentials to various parts of the myocardium. Smooth muscle cells possess a contractile apparatus of thin and thick filaments and a cytoskeleton of desmin and vimentin intermediate filaments. Thin filaments contain actin, tropomyosin (a smooth muscle isoform), caldesmon, and calponin. Thin filaments are attached to cytoplasmic densities or dense bodies, which contain -actinin and are located throughout the sarcoplasm and close to the sarcolemma. Contraction of smooth muscle is triggered by a variety of impulses, including mechanical (passive stretching), electrical (depolarization at nerve endings), and chemical (hormones acting by a second messenger) stimuli. Because smooth muscle cells lack T tubules, Ca2 is delivered by caveolae and cytoplasmic vesicles. Early in development, these cells express MyoD transcription factor, which plays a key role in activation of muscle-specific gene expressions and differentiation of all skeletal muscle lineages. Repair of skeletal muscle and its regeneration can occur from multipotential myogenic stem cells called satellite cells. These cells are left over from fetal development and express Pax7 transcription factor. They co-express Pax7 with MyoD to become myogenic precursors of skeletal muscle cells. Smooth muscle cells are capable of dividing to maintain or increase their number and size. Two major types are recognized: striated muscle, in which the cells exhibit a cross-striation pattern when observed at the light microscope level; and smooth muscle, in which the cells lack striations. Striated muscle is further subclassified based on location, namely, skeletal muscle, visceral striated muscle, and cardiac muscle. Skeletal muscle is attached to bone and is responsible for movement of the axial and appendicular skeleton and for maintenance of body position and posture. Visceral striated muscle is morphologically identical but is restricted to soft tissues, including the tongue, pharynx, upper part of the esophagus, and the diaphragm. Cardiac muscle is a type of striated muscle found in the heart and the base of the large veins that empty into the heart. The skeletal and visceral striated muscle cells, more commonly called fibers, are a multinucleated syncytium formed during development by the fusion of individual small muscle cells called myoblasts. Surrounding each fiber is a delicate mesh of collagen fibrils referred to as endomysium. In turn, bundles of muscle fibers that form functional units within a muscle are surrounded by a thicker connective tissue layer. Lastly, a sheath of dense connective tissue that surrounds the muscle is referred to as epimysium.
Diseases
- Senter syndrome
- Lambert Eaton myasthenic syndrome (Lambert Eaton paraneoplastic cerebellar degeneration)
- Cerebelloparenchymal disorder 3
- Spondylarthropathy
- Dermatographic urticaria
- Eosinophilic fasciitis
- Renier Gabreels Jasper syndrome
- Gordon hyperkaliemia-hypertension syndrome
- Alternating hemiplegia of childhood
- Hemoglobinopathy
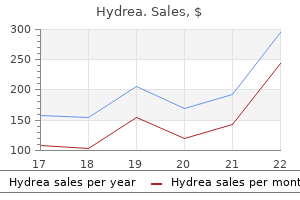
Effective hydrea 500 mg
Crystalline inclusions contained in certain cells are recognized in the light microscope treatment 4 sore throat buy hydrea 500 mg fast delivery. In humans, such inclusions are found in the Sertoli (sustentacular) and Leydig (interstitial) cells of the testis. Although some of these inclusions contain viral proteins, storage material, or cellular metabolites, the significance of others is not clear. This network provides a structural substratum on which cytoplasmic reactions occur, such as those involving free ribosomes, and along which regulated and directed cytoplasmic transport and movement of organelles occur. Cells have two major compartments: the cytoplasm (contains organelles and inclusions surrounded by cytoplasmic matrix) and the nucleus (contains genome). Organelles are metabolically active complexes or compartments that are classified into membranous and nonmembranous organelles. It is composed of phospholipids, cholesterol, embedded integral membrane proteins, and associated peripheral membrane proteins. Integral membrane proteins have important functions in cell metabolism, regulation, and integration. They include pumps, channels, receptor proteins, linker proteins, enzymes, and structural proteins. Lipid rafts represent microdomains in the plasma membrane that contain high concentrations of cholesterol and glycosphingolipids. They are movable signaling platforms that carry integral and peripheral membrane proteins. Vesicle budding permits molecules to enter the cell (endocytosis), leave the cell (exocytosis), or travel within the cell cytoplasm in transport vesicles. It is dependent on three different mechanisms: pinocytosis (uptake of fluids and dissolved small proteins), phagocytosis (uptake of large particles), and receptor-mediated endocytosis (uptake of specific molecules that bind to receptors). Exocytosis is the process of cellular secretion in which transport vesicles, when fused with plasma membrane, discharge their content into the extracellular space. In constitutive exocytosis, the content of transport vesicles is continuously delivered and discharged at the plasma membrane. In regulated secretory exocytosis, the content of vesicles is stored within the cell and released pending hormonal or neural stimulation. Lysosomes are digestive organelles containing hydrolytic enzymes that degrade substances derived from endocytosis and from the cell itself (autophagy). They have a unique membrane made of specific structural proteins resistant to hydrolytic digestion. Lysosomes develop from endosomes by receiving newly synthesized lysosomal proteins (enzymes and structural proteins) that are targeted via the mannose-6-phosphate (M-6-P) lysosomal targeting signals. Proteasomes are nonmembranous organelles that also function in degradation of proteins. They represent cytoplasmic protein complexes that destroy damaged (misfolded) or unwanted proteins that have been labeled for destruction with ubiquitin without the involvement of lysosomes. It is the site of protein synthesis and posttranslational modification of newly synthesized proteins. It contains detoxifying enzymes (liver) and enzymes for glycogen and lipid metabolism. The Golgi apparatus represents a series of stacked, flattened cisternae and functions in the posttranslational modification, sorting, and packaging of proteins directed to four major cellular destinations: apical and basolateral plasma membrane, endosomes and lysosomes, and apical cytoplasm (for storage and/or secretion). They are abundant in cells that generate and expend large amounts of energy, and they regulate apoptosis (programmed cell death). Peroxisomes are small organelles involved in the production and degradation of H2O2 and in the degradation of fatty acids. Microtubules form tracts for intracellular vesicular transport and mitotic spindles; they are also responsible for the movement of cilia and flagella and for the maintenance of cell shape. Movement of intracellular organelles along microtubules is generated by molecular motor proteins (dyneins and kinesins). Actin filaments (microfilaments) are thinner (6 to 8 nm in diameter), shorter, and more flexible than microtubules. They are composed of polymerized G-actin (globulin actin) molecules that form F-actin (filamentous actin). Actin filaments are also responsible for cell-to-extracellular matrix attachment (focal adhesions), movement of membrane proteins, formation of the structural core of microvilli, and cell motility through the creation of cell extensions (lamellipodia and filopodia). Intermediate filaments are rope-like filaments (8 to 10 nm in diameter) that add stability to the cell and interact with cell junctions (desmosomes and hemidesmosomes). Intermediate filaments are formed from nonpolar and highly variable intermediate filament subunits that include keratins (found in epithelial cells), vimentin (mesodermally derived cells), desmin (muscle cells), neurofilament proteins (nerve cells), lamins (nucleus), and beaded filament proteins (eye lens). Centrioles are paired, short, rod-like cytoplasmic cylinders built from nine microtubule triplets. The nuclear envelope is a double membrane system that surrounds the nucleus of the cell. It consists of an inner and an outer membrane separated by a perinuclear cisternal space and perforated by nuclear pores. A simple microscopic evaluation of the nucleus provides a great deal of information about cell well-being. This is accomplished by the formation of a unique nucleoprotein complex called chromatin. Further folding of chromatin, such as that which occurs during mitosis, produces structures called chromosomes. Chromatin proteins include five basic proteins called histones along with other nonhistone proteins. The nuclear wall consists of a double membrane envelope that surrounds the nucleus. The inner membrane is adjacent to nuclear intermediate filaments that form the nuclear lamina. This electron micrograph, prepared by the quick-freeze deep-etch technique, shows the nucleus, the large spherical object, surrounded by the nuclear envelope. Sequencing of the human genome took about 13 years and was successfully completed in 2003 by the Human Genome Project. For years, it was thought that genes were usually present in two copies in a genome. For instance, genes that were thought to always occur in two copies per genome have sometimes one, three, or more copies. In general, two forms of chromatin are found in the nucleus: a condensed form called heterochromatin and a dispersed form called euchromatin. There are two recognizable types of heterochromatin: constitutive and facultative. Large amounts of constitutive heterochromatin are found in chromosomes near the centromeres and telomeres. Facultative heterochromatin is also condensed and is not involved in the transcription process. In contrast to constitutive heterochromatin, facultative heterochromatin is not repetitive and has inconsistent nuclear and chromosomal localization when compared with other cell types. Facultative heterochromatin may undergo active transcription in certain cells (see Barr body description on page 78) due to specific conditions such as explicit cell cycle stages, nuclear localization changes. The densely staining material is highly condensed chromatin called heterochromatin, and the lightly staining material (where most transcribed genes are located) is a dispersed form called euchromatin. It is the phosphate groups of the chromatin nucleus (the structure light microscopists formerly referred to as the nuclear membrane actually consists largely of marginal chromatin). Karyosomes are discrete bodies of chromatin irregular in size and shape that are found throughout the nucleus. Nucleolar-associated chromatin is chromatin found in association with the nucleolus. The nucleus of this active cell, exclusive of the nucleoli, comprises almost entirely extended chromatin or euchromatin. The smaller nucleus belongs to a circulating lymphocyte (the entire cell is shown in the micrograph). It is the heterochromatin that accounts for the conspicuous staining of the nucleus in hematoxylin and eosin (H&E) preparations. It is present within the nucleoplasm in the "clear" areas between and around the heterochromatin. In routine electron micrographs, there is no sharp delineation between euchromatin and heterochromatin; both have a granular, filamentous appearance, but the euchromatin is less tightly packed. Heterochromatin predominates in metabolically inactive cells such as small circulating lymphocytes and sperm or in cells that produce one major product such as plasma cells.
Order generic hydrea on line
After entry into the vascular system from the bone marrow symptoms gallstones cheap 500 mg hydrea fast delivery, the platelets circulate as discoid structures about 2 to 3 m in diameter. Structurally, platelets may be divided into four zones based on organization and function. The glycocalyx consists of glycoproteins, glycosaminoglycans, and several coagulation factors adsorbed from the plasma. The structural zone, near the periphery, comprises microtubules, actin filaments, myosin, and actin-binding proteins that form a network supporting the plasma membrane. The marginal band containing 8 to 24 coiled microtubules resides as a bundle immediately below the actin filament network. It consists of mitochondria, peroxisomes, glycogen particles, and at least three types of granules dispersed within the cytoplasm. The most numerous granules are granules (300 to 500 nm in diameter) that contain mainly fibrinogen, coagulation factors, plasminogen, plasminogen activator inhibitor, and platelet-derived growth factor. The contents of these granules play an important role in the initial phase of vessel repair, blood coagulation, and platelet aggregation. High-magnification electron micrograph of a platelet situated between an erythrocyte on the left and an endothelial cell on the right. Visible organelles include a mitochondrion, microtubules, a single profile of the surfaceconnected open canalicular system, profiles of the dense tubular system, the moderately dense granules, a single very dense granule, and glycogen particles. They facilitate platelet adhesion and vasoconstriction in the area of the injured vessel. The granules are similar to lysosomes found in other cells and contain several hydrolytic enzymes. The contents of granules function in clot resorption during the later stages of vessel repair. In effect, open canaliculi are invaginations into the cytoplasm from the plasma membrane. They continuously survey the endothelial lining of blood vessels for gaps and breaks. When a blood vessel wall is injured or broken, the exposed connective tissue at the damaged site promotes platelet adhesion. Serotonin is a potent vasoconstrictor that causes the vascular smooth muscle cells to contract, thereby reducing local blood flow at the site of injury. The glycocalyx of the platelets provides a reaction surface for the conversion of soluble fibrinogen into fibrin. The initial platelet plug is transformed into a definitive clot known as a secondary hemostatic plug by additional tissue factors secreted by the damaged blood vessel. After the definitive clot is formed, platelets cause clot retraction, probably as a function of the actin and myosin found in the structural zone of the platelet. Contraction of the clot permits the return of normal blood flow through the vessel. Finally, after the clot has served its function, it is lysed by plasmin, a fibrinolytic enzyme that circulates in the plasma in an inactive form known as plasminogen. High-magnification scanning electron micrograph shows initial stage of blood clot formation. Red blood cells are entrapped in a loose mesh of fibrin fibers that are extensively cross-linked to form an impermeable hemostatic plug that prevents movement of cells and fluids from the lumen of the injured vessel. An additional role of platelets is to help repair the injured tissues beyond the vessel itself. Platelet-derived growth factor released from the granules stimulates smooth muscle cells and fibroblasts to divide and allow tissue repair. It provides relative numbers and calculations obtained from the cells (erythrocytes and leukocytes) and formed elements (thrombocytes) in the blood sample. These calculations are usually performed by automated blood cell counters that analyze different components of blood using the principle of flow cytometry design. As a thin stream of fluid with suspended cells flows through narrow tubing in the cell counter, the light detector and electrical impedance sensor identify different cell types based on their size and electrical resistance. Data obtained from automatic blood analyzers were usually very accurate due to the large number of cells counted (10,000) in each category. However, in some cases, manual cell count under a light microscope is still necessary. Leukocyte count can also be elevated after strenuous exercise because of stress, or in pregnancy and labor. Hyperleukocytosis (leukocyte count 100 109 cells/L) is commonly an indication of leukemia (type of blood cancer). The major types of white blood cells reported are neutrophils, eosinophils, basophils, lymphocytes, and monocytes. Each type of these cells plays a different role in protecting the body, and percentages of their distribution in the blood sample give important information about the status of the immune system. Refer to the appropriate sections of this chapter for descriptions and functions of these cells. Elevated erythrocyte count (polycythemia) may be related to intrinsic factors affecting erythrocyte production in the bone marrow (primary polycythemia) or as a response to stimuli. Secondary polycythemia is usually due to increased production of erythropoietin in response to chronic hypoxia, high altitude, or an erythropoietinsecreting tumor. Decreased erythrocyte count (anemia) is caused by loss of blood (external or internal bleeding), iron or vitamin B12 deficiencies, poor nutrition, pregnancy, chronic diseases, and genetic disorders. Normal Hgb values are 14 to 18 g/dL (140 to 180 g/L) in males and 12 to 15 g/dL (120 to 150 g/L) in females. Hematocrit and hemoglobin values are the two major tests that show if anemia or polycythemia is present. These indices are automatically calculated from other measurements and are useful in differential diagnosis. Thrombocytes are important in blood clotting, and their elevation (thrombocythemia) may be related to proliferative disorders of the bone marrow, inflammation, decreased function of spleen, or as a result of splenectomy. Low thrombocyte count (thrombocytopenia) may be related to decreased production of thrombocytes in bone marrow. Blood cells have a limited life span; they are continuously produced and destroyed. The ultimate objective of hemopoiesis is to maintain a constant level of the different cell types found in the peripheral blood. Both the human erythrocyte (life span of 120 days) and the platelet (life span of 10 days) spend their entire life in the circulating blood. Leukocytes, however, migrate out of the circulation shortly after entering it from the bone marrow and spend most of their variable life spans (and perform all of their functions) in the tissues. In the adult, erythrocytes, granulocytes, monocytes, and platelets are formed in the red bone marrow; lymphocytes are also formed in the red bone marrow and in the lymphatic tissues. To study the stages of blood cell formation, a sample of bone marrow aspirate (see page 302) is prepared as a stained smear in a manner similar to that of a smear of blood. During fetal life, both erythrocytes and leukocytes are formed in several organs before the differentiation of the bone marrow. The first or yolk-sac phase of hemopoiesis begins in the third week of gestation and is characterized by the formation of "blood islands" in the wall of the yolk sac of the embryo. Blood cell formation in these sites is largely limited to erythroid cells, although some leukopoiesis occurs in the liver. The liver is the major blood-forming organ in the fetus during the second trimester. The third or bone marrow phase of fetal hemopoiesis and leukopoiesis involves the bone marrow (and other lymphatic tissues) and begins during the second trimester of pregnancy. Cytokines (including hemopoietic growth factors) may and do act individually and severally at any point in the process from the first stem cell to the mature blood or connective tissue cell. If committed to enter the mast cell lineage, the basophil/mast cell progenitor cell migrates to the spleen where it differentiates into a mast cell progenitor cell. After further differentiation in the spleen, it migrates to the intestine to become a mast cell precursor. Although it is difficult to discern, these cells are located between developing liver cells and the wall of the vascular sinus.

Order hydrea once a day
In both sections and smears medications harmful to kidneys hydrea 500mg online, many cells must be examined to find those whose orientation is suitable for the display of the Barr body. The second X chromosome of the female patient is repressed in the interphase nucleus and can be demonstrated in the neutrophil as a drumstick-appearing appendage (arrow) on a nuclear lobe. Granular material (pars granulosa) represents the site of initial ribosomal assembly and contains densely packed preribosomal particles. The network formed by the granular and the fibrillar materials is called the nucleolonema. The nucleolus varies in size but is particularly well developed in cells active in protein synthesis. Chromosome analysis can be performed on peripheral blood, bone marrow, tissues (such as skin or chorionic villi obtained from biopsies), and cells obtained from amniotic fluid during amniocentesis. Studies of chromosomes begin with the extraction of whole chromosomes from the nuclei of dividing cells. To obtain an image of all of the chromosomes, a mixture of different probes is used to produce different colors in each chromosome. Karyotypes labeled by this method allow cytogeneticists to perform a comprehensive analysis of changes in the number of chromosomes and chromosomal abnormalities such as additions or deletions. It is clearly visible on this color image that a part of the original chromosome 8 (aqua blue region) is now attached to chromosome 14, and a small portion of chromosome 14 (red region) is now part of chromosome 8. Note that one homolog of chromosome 15 has lost that region (no orange signal is visible). When the deletion is inherited from the mother, patients develop Angelman syndrome; when inherited from the father, patients develop Prader-Willi syndrome. The partially assembled ribosomal subunits (preribosomes) are exported from the nucleus via nuclear pores for full assembly into mature ribosomes in the cytoplasm. Nucleostemin is a newly identified protein that has been the Cell Nucleus found within the nucleolus. Nucleostemin is a p53-binding protein that regulates the cell cycle and influences cell differentiation (page 85). The presence of nucleostemin in malignant cells suggests that it could play a role in their uncontrolled proliferation (Folder 3. These viruses can use components of the nucleolus as part of their own replication process. Evidence suggests that viruses may target the nucleolus and its components to favor viral transcription and translation and perhaps alter the cell cycle to promote viral replication. The nucleolus stains intensely with hematoxylin and basic dyes and metachromatically with thionine dyes. The nuclear envelope is assembled from two (outer and inner) nuclear membranes with a perinuclear cisternal space between them. Polyribosomes are often attached to ribosomal docking proteins present on the cytoplasmic side of the outer nuclear membrane. In addition, the inner nuclear membrane contains specific lamin receptors and several lamina-associated proteins that bind to chromosomes and secure the attachment of the nuclear lamina. The nuclear lamina is formed by intermediate filaments and lies adjacent to the inner nuclear membrane. Thus, when examined in the light microscope, nucleoli appear Feulgen-negative with Feulgen-positive nucleolus-associated chromatin that often rims the nucleolus. Nuclear Envelope the nuclear envelope, formed by two membranes with a perinuclear cisternal space between them, separates the nucleoplasm from the cytoplasm. The nuclear envelope provides a selectively permeable membranous barrier between the nuclear compartment and the nuclear lamina, a thin, electron-dense intermediate filament network-like layer, resides underneath the nuclear membrane. If the membranous component of the nuclear envelope is disrupted by exposure to detergent, the nuclear lamina remains, and the nucleus retains its shape. The major components of the lamina, as determined by biochemical isolation, are nuclear lamins, a specialized type of nuclear intermediate filament (see page 63), and lamin-associated proteins. Nuclear lamina is essentially composed of lamin A and lamin C proteins that form intermediate filaments. For instance, inactivation of tumor-suppressor genes has been shown to play a role in the growth and division of cancer cells. By screening patients for mutations in these genes, much earlier detection of cancer can be accomplished. It is also now known why in some individuals, p53 mutations make their tumors resistant to radiotherapy. However, these cells will not die because of the absence of functional p53, which triggers apoptosis. This schematic drawing shows the structure of the nuclear lamina adjacent to the inner nuclear membrane. Note that the nuclear envelope is pierced by nuclear pore complexes, which allow for selective bidirectional transport of molecules between nucleus and cytoplasm. It is formed by intermediate filaments (lamins) that are arranged in a square lattice. Unlike other cytoplasmic intermediate filaments, lamins disassemble during mitosis and reassemble when mitosis ends. The nuclear lamina appears to serve as scaffolding for chromatin, chromatin-associated proteins, nuclear pores, and the membranes of the nuclear envelope. In addition, it is involved in nuclear organization, cell-cycle regulation, differentiation, and gene expression. Impairment in nuclear lamina architecture or function is associated with certain genetic diseases (laminopathies) and apoptosis. Mutations in lamin A/C cause tissue-specific diseases that affect striated muscle, adipose tissue, peripheral nerve or skeletal development, and premature aging. At numerous sites, the paired membranes of the nuclear envelope are punctuated by 70- to 80-nm "openings" through the envelope. These nuclear pores are formed from the merging of the inner and outer membranes of the nuclear envelope. Because such profiles are thought to represent either ribosomes or other protein complexes (transporters) captured during their passage through the pore at the time of fixation, the term central plug/transporter is commonly used to describe this feature. Conversely, nuclear proteins, such as histones and lamins, are produced in the cytoplasm and are transported through nuclear pores into the nucleus. The effective size of the pore is about 9 nm for substances that cross by diffusion rather than the 70- to 80-nm measurement of the pore boundary. However, even the smaller nuclear proteins that are capable of diffusion are selectively transported, presumably because the rate is faster than simple diffusion. The Cell Nucleus During cell division, the nuclear envelope is disassembled to allow chromosome separation and is later reassembled as the daughter cells form. Note the visible nuclear pore complexes (arrows) and the two membranes that constitute the nuclear envelope. At the periphery of each pore, the outer and inner membranes of the nuclear envelope appear continuous. In late prophase of cell division, enzymes (kinases) are activated that cause phosphorylation of the nuclear lamins and other lamina-associated proteins of the nuclear envelope. After phosphorylation, the proteins become soluble, and the nuclear envelope disassembles. The lipid component of the nuclear membranes then disassociates from the proteins and is retained in small cytoplasmic vesicles. The replicated chromosomes then attach to the microtubules of the mitotic spindle and undergo active movement. Reassembly of the nuclear envelope begins in late anaphase, when phosphatases are activated to remove the phosphate residues from the nuclear lamins. During telophase, the nuclear lamins begin to repolymerize and form the nuclear lamina material around each set of daughter chromosomes. At the same time, vesicles containing the lipid components of the nuclear membranes and structural membrane protein components fuse, and an envelope is formed on the surface of the alreadyreassembled nuclear lamina. By the end of telophase, formation of a nuclear envelope in each daughter cell is complete. Ribosomal proteins are partially assembled into ribosomal subunits in the nucleolus and are transported through nuclear pores to the Nucleoplasm Nucleoplasm is the material enclosed by the nuclear envelope exclusive of the chromatin and the nucleolus. They protrude from the cytoplasmic ring subunits and point toward the center of the structure. It must be assumed, however, that many proteins and other metabolites reside in or pass through the nucleus in relation to the synthetic and metabolic activity of the chromatin and nucleolus.
Satureja calamintha (Calamint). Hydrea.
- Dosing considerations for Calamint.
- What is Calamint?
- Colds, fever, breathing problems, and chest congestion.
- Are there safety concerns?
- How does Calamint work?
Source: http://www.rxlist.com/script/main/art.asp?articlekey=96608

Order hydrea canada
Because of the presence of the sulfate groups symptoms exhaustion hydrea 500mg overnight delivery, aggrecan molecules have a large negative charge with an affinity for water molecules. Each linear hyaluronan molecule is associated with a large number of aggrecan molecules (more than 300), which are bound to the hyaluronan by link proteins at the N terminus of the molecule to form large proteoglycan aggregates. The entrapment of these aggregates within the intricate matrix of collagen fibrils is responsible for the unique biomechanical properties of hyaline cartilage. These proteoglycans do not form aggregates but bind to other molecules and help stabilize the matrix. Multiadhesive glycoproteins, also referred to as noncollagenous and nonproteoglycan-linked glycoproteins, influence proteoglycan monomer (aggrecan) interactions between the chondrocytes and the matrix molecules. Multiadhesive glycoproteins have clinical value as markers of cartilage turnover and degeneration. Hyaline cartilage matrix is highly hydrated to provide resilience and diffusion of small metabolites. These large hydrodynamic domains in the matrix are accountable for imparting resilience to the cartilage. Some of the water is bound loosely enough to allow diffusion of small metabolites to and from the chondrocytes. In articular cartilage, both transient and regional changes occur in water content during joint movement and when the joint is subjected to pressure. A hyaluronan molecule forming a linear aggregate with many proteoglycan monomers is interwoven with a network of collagen fibrils. The proteoglycan monomer (such as aggrecan) consists of approximately 180 glycosaminoglycans joined to a core protein. The end of the core protein contains a hyaluronan-binding region that is joined to the hyaluronan by a link protein. Throughout life, cartilage undergoes continuous internal remodeling as the cells replace matrix molecules lost through degradation. Normal matrix turnover depends on the ability of the chondrocytes to detect changes in matrix composition. In addition, the matrix acts as a signal transducer for the embedded chondrocytes. Thus, pressure loads applied to the cartilage, as in synovial joints, create mechanical, electrical, and chemical signals that help direct the synthetic activity of the chondrocytes. As the body ages, however, the composition of the matrix changes, and the chondrocytes lose their ability to respond to these stimuli. Chondrocytes are specialized cells that produce and maintain the extracellular matrix. This specimen was preserved in glutaraldehyde, embedded in plastic, and stained with H&E. The chondrocytes, especially those in the upper part of the photomicrograph, are well preserved. The cytoplasm is deeply stained, exhibiting a distinct and relatively homogeneous basophilia. This layer represents deposition of new cartilage (appositional growth) on the surface of the existing hyaline cartilage. Mature chondrocytes with clearly visible nuclei (N) reside in the lacunae and are well preserved in this specimen. Growth from within the cartilage (interstitial growth) is reflected by the chondrocyte pairs and clusters that are responsible for the formation of isogenous groups (rectangles). When the chondrocytes are present in isogenous groups, they represent cells that have recently divided. As the newly divided chondrocytes produce the matrix material that surrounds them, they are dispersed. They also secrete metalloproteinases, enzymes that degrade cartilage matrix, allowing the cells to expand and reposition themselves within the growing isogenous group. Chondrocytes not only secrete the collagen present in the matrix but also all of the glycosaminoglycans and proteoglycans. In older, less active cells, the Golgi apparatus is smaller; clear areas of cytoplasm, when evident, usually indicate sites of extracted lipid droplets and glycogen stores. In such specimens, chondrocytes also display considerable distortion resulting from shrinkage after the glycogen and lipid are lost during preparation of the tissue. Thus, the basophilia and metachromasia seen in stained sections of cartilage provide information about the distribution and relative concentration of sulfated proteoglycans. The territorial matrix is a region that is more removed from the immediate vicinity of the chondrocytes. It also has a lower concentration of sulfated proteoglycans and stains less intensely than the capsular matrix. The interterritorial matrix is a region that surrounds the territorial matrix and occupies the space between groups of chondrocytes. G M In addition to these regional differences in the concentration of sulfated proteoglycans and distribution of collagen fibrils, there is a decrease in proteoglycan content that occurs as cartilage ages, which is also reflected by staining differences. The particularly large particles adjacent to the cell are located in the region of the matrix that is identified as the capsule or territorial matrix. Initially, most long bones are represented by cartilage models that resemble the shape of the mature bone (Plate 8, page 208). During the developmental process, in which most of the cartilage is replaced by bone, residual cartilage at the proximal and distal end of the bone serves as growth sites called epiphyseal growth plates (epiphyseal discs). The hyaline cartilage of developing tarsal bones will be replaced by bone as endochondral ossification proceeds. In this early stage of development, synovial joints are being formed between developing tarsal bones. Note that nonarticulating surfaces of the hyaline cartilage models of tarsal bones are covered by perichondrium, which also contributes to the development of joint capsules. Also, a developing tendon (T) is evident in the indentation of the cartilage seen on the left side of the micrograph. A disc of hyaline cartilage-the epiphyseal plate- separates the more proximally located epiphysis from the funnel-shaped diaphysis located distal to the plate. The articular cartilage on the surface of the epiphysis contributes to the synovial joint and is also composed of hyaline cartilage. The cartilage of the epiphyseal plate disappears when lengthwise growth of the bone is completed, but the articular cartilage remains throughout life. Hyaline cartilage also exists in the adult as the skeletal unit in the trachea, bronchi, larynx, and nose. A firmly attached connective tissue, the perichondrium, surrounds hyaline cartilage. Hyaline cartilage that covers the articular surfaces of movable joints is termed articular cartilage. In general, the the perichondrium is a dense irregular connective tissue composed of cells that are indistinguishable from fibroblasts. In many respects, the perichondrium resembles the capsule that surrounds glands and many organs. When actively growing, the perichondrium appears divided into an inner cellular layer, which gives rise to new cartilage cells, and an outer fibrous layer. This division is not always evident, especially in perichondrium that is not actively producing new cartilage or in very slow-growing cartilage. Also, on the opposite surface, the cartilage contacts the bone, and there is no perichondrium. Articular cartilage is a remnant of the original hyaline cartilage template of the developing bone, and it persists throughout adult life. This diagram shows the organization of the collagen network and chondrocytes in the various zones of articular cartilage. Also, this zone is lighter staining than the matrix of the more superficial zones. Collagen fibrils are less organized and are arranged in a somewhat oblique orientation to the surface. The deep (radial) zone is characterized by small, round chondrocytes that are arranged in short columns perpendicular to the free surface of the cartilage.
Order genuine hydrea
Salivation is part of a reflex arc that is normally stimulated by the ingestion of food medicine hat college order hydrea with mastercard, although sight, smell, or even thoughts of food can also stimulate salivation. Thus, the acinus is rather large, and much of it is usually not seen within the plane of a single section. The serous component of the gland is composed largely of demilunes, but occasional serous acini are present. As noted earlier, some of the serous demilunes may be sectioned in a plane that does not include the mucous component of the acinus, thus giving the appearance of a serous acinus. The ducts of the sublingual gland that are observed most frequently in a section are the intralobular ducts. They are the equivalent of the striated duct of the submandibular and parotid glands but lack the extensive basal infoldings and mitochondrial array that creates the striations. The area within the rectangle includes part of this duct and is shown at higher magnification in the figure below. The juncture between the acinus and the beginning of the intercalated duct is marked by an arrowhead. The intercalated duct is composed of a flattened or low columnar epithelium similar to that seen in the other salivary glands. The intercalated ducts of the sublingual gland are extremely short, however, and thus are usually difficult to find. The intercalated duct seen in this micrograph joins with one or more other intercalated ducts to become the intralobular duct (InD), which is identified by its columnar epithelium and relatively large lumen. The point of transition from intercalated to intralobular duct is not recognizable in the micrograph, however, because the duct wall has only been grazed and the shape of the cells cannot be determined. The area selected for this higher magnification also reveals isolated cell clusters that bear some resemblance to serous acini. It is likely, however, that these cells are actually mucous cells that either have been cut in a plane parallel to their base and do not include the mucinogen-containing portions of the cell or are in a state of activity in which, after depletion of their granules, the production of new mucinogen granules does not yet suffice to give the characteristic "empty" mucous cell appearance. An additional important feature of the connective tissue stroma is the presence of numerous lymphocytes and plasma cells. The plasma cells are associated with the production of salivary IgA and are also present in the other salivary glands. An adventitia consisting only of connective tissue is found where the wall of the tube is directly attached or fixed to adjoining structures. This composite diagram shows the wall structure of the alimentary canal in four representative organs: esophagus, stomach, small intestine, and large intestine. Note that villi, a characteristic feature of the small intestine, are not present in other parts of the alimentary canal. Mucosal glands are present throughout the length of the alimentary canal but sparingly in the esophagus and oral cavity. The extramural glands (liver and pancreas) empty into the duodenum (first part of the small intestine). Diffuse lymphatic tissues and nodules are found in the lamina propria throughout the entire length of the alimentary canal (shown here only in the large intestine). Nerves, blood vessels, and lymphatic vessels reach the alimentary canal via the mesenteries or via adjacent connective tissue (tunica adventitia as in the retroperitoneal organs). The epithelium differs throughout the alimentary canal and is adapted to the specific function of each part of the tube. The histologic characteristics of these layers and their functions are described below in relation to specific regions of the digestive tube. The epithelium of the mucosa serves as a barrier that separates the lumen of the alimentary canal from the rest of the organism. The absorptive function of the mucosa allows the movement of digested nutrients, water, and electrolytes into the blood and lymph vessels. The absorption of digested nutrients, water, and electrolytes is possible because of projections of the mucosa and submucosa into the lumen of the digestive tract. These surface projections greatly increase the surface area available for absorption and vary in size and orientation. The barrier aids in protection of the individual from the entry of antigens, pathogens, and other noxious substances. In the esophagus, a stratified squamous epithelium provides protection from physical abrasion by ingested food. In the gastrointestinal portion of the alimentary tract, tight junctions between the simple columnar epithelial cells of the mucosa serve as a selectively permeable barrier. Most epithelial cells transport products of digestion and other essential substances such as water through the cell and into the extracellular space beneath the tight junctions. Villi are mucosal projections that cover the entire surface of the small intestine, the principal site of absorption of the products of digestion. Microvilli are tightly packed, microscopic projections of the apical surface of intestinal absorptive cells. It provides additional surface for adsorption and includes enzymes secreted by the absorptive cells that are essential for the final steps of digestion of proteins and sugars. The epithelium selectively absorbs the products of digestion both for its own cells and for transport into the vascular system for distribution to other tissues. The secretory function of the mucosa provides lubrication and delivers digestive enzymes, hormones, and antibodies into the lumen of the alimentary tube. They tend to be located on the side of the tube opposite the attachment of the mesentery. Secretion is carried out largely by glands distributed through- out the length of the digestive tube. The mucosal epithelium also secretes antibodies that it receives from the underlying connective tissue. Contraction of this muscle produces movement of the mucosa, forming ridges and valleys that facilitate absorption and secretion. This localized movement of the mucosa is independent of the peristaltic movement of the entire wall of the digestive tract. The liver and the pancreas are extramural digestive glands (see Chapter 18) that greatly increase the secretory capacity of the digestive system. They deliver their secretions into the duodenum, the first part of the small intestine. The lamina propria contains glands, vessels that transport absorbed substances, and components of the immune system. In general, they lubricate the epithelial surface to protect the mucosa from mechanical and chemical injury. These glands are described below in relation to specific regions of the digestive tube. In segments of the digestive tract in which absorption occurs, principally the small and large intestines, the absorbed products of digestion diffuse into the blood and lymphatic vessels of the lamina propria for distribution. Typically, the blood capillaries are of the fenestrated type and collect most of the absorbed metabolites. In the small intestine, lymphatic capillaries are numerous and receive some absorbed lipids and proteins. The lymphatic tissues in the lamina propria function as an integrated immunologic barrier that protects against pathogens and other antigenic substances that could potentially enter through the mucosa from the lumen of the alimentary canal. The extensive nerve network in the submucosa contains visceral sensory fibers mainly of sympathetic origin, parasympathetic (terminal) ganglia, and preganglionic and postganglionic parasympathetic nerve fibers. The nerve cell bodies of parasympathetic ganglia and their postganglionic nerve fibers represent the enteric nervous system, the third division of the autonomic nervous system. This system is primarily responsible for innervating the smooth muscle layers of the alimentary canal and can function totally independently of the central nervous system. For example, they are present in the esophagus and the initial portion of the duodenum. In histologic sections, the presence of these glands often aids in identifying the specific segment or region of the tract. Muscularis Externa In most parts of the digestive tract, the muscularis externa consists of two concentric and relatively thick layers of smooth muscle.
Hydrea 500 mg low cost
Peristalsis is marked by constriction and shortening of the tube medicine pills discount hydrea 500 mg mastercard, which moves the contents through the intestinal tract. A few sites along the digestive tube exhibit variations in the muscularis externa. For example, in the wall of the proximal portion of the esophagus (pharyngoesophageal sphincter) and around the anal canal (external anal sphincter), striated muscle forms part of the muscularis externa. In the stomach, a third, obliquely oriented layer of smooth muscle is present deep into the circular layer. Finally, in the large intestine, part of the longitudinal smooth muscle layer is thickened to form three distinct, equally spaced longitudinal bands called teniae coli. During contraction, the teniae facilitate shortening of the tube to move its contents. The circular smooth muscle layer forms sphincters at specific locations along the digestive tract. Large blood and lymphatic vessels and nerve trunks travel through the serosa (from and to the mesentery) to reach the wall of the digestive tract. Large amounts of adipose tissue can develop in the connective tissue of the serosa (and in the mesentery). These include the thoracic part of the esophagus and portions of structures in the abdominal and pelvic cavities that are fixed to the cavity wall-the duodenum, ascending and descending colon, rectum, and anal canal. These structures are attached to the abdominal and pelvic wall by connective tissue, the adventitia, which blends with the connective tissue of the wall. At several points along the digestive tract, the circular muscle layer is thickened to form sphincters or valves. As the name implies, this sphincter is located at the lower end of the esophagus; its action is reinforced by the diaphragm that surrounds this part of the esophagus as it passes into the abdominal cavity. It creates a pressure difference between the esophagus and stomach that prevents reflux of gastric contents into the esophagus. Abnormal relaxation of this sphincter allows acidic content of the stomach to return (reflux) into the esophagus. Located at the junction of the pylorus of the stomach and duodenum (gastroduodenal sphincter), it controls the release of chyme, the partially digested contents of the stomach, into the duodenum. Located at the junction of the small and large intestines, it prevents reflux of the contents of the colon with its high bacterial count into the distal ileum, which normally has a low bacterial count. This, the most distally located sphincter, surrounds the anal canal and prevents passage of the feces into the anal canal from the undistended rectum. Serosa and Adventitia Serosa or adventitia constitutes the outermost layer of the alimentary canal. The serosa is a serous membrane consisting of a layer of simple squamous epithelium, called the mesothelium, and a small amount of underlying connective tissue. The serosa is the most superficial layer of those parts of the digestive tract that are suspended in the peritoneal cavity. As such, the serosa is continuous with both the mesentery and the lining of the abdominal cavity. The esophagus courses through the neck and mediastinum, where it is attached to adjacent structures by connective tissue. As it enters the abdominal cavity, it is free for a short distance, approximately 1 to 2 cm. When a bolus of food passes through the esophagus, the lumen expands without mucosal injury. In many animals, however, the epithelium is keratinized, reflecting a coarse food diet. In humans, the surface cells may exhibit some keratohyalin granules, but keratinization does not normally occur. The underlying lamina propria is similar to the lamina propria throughout the alimentary tract; diffuse lymphatic tissue is scattered throughout, and lymphatic nodules are present, often in proximity to ducts of the esophageal mucous glands (see page 573). The deep layer of the mucosa, the muscularis mucosae, is composed of longitudinally organized smooth muscle that begins near the level of the cricoid cartilage. It is unusually thick in the proximal portion of the esophagus and presumably functions as an aid in swallowing. The submucosa consists of dense irregular connective tissue that contains the larger blood and lymphatic vessels, nerve fibers, and ganglion cells. In addition, diffuse lymphatic tissue and lymphatic nodules are present mostly in the upper and lower parts of the esophagus where submucosal glands are more prevalent. The muscularis externa consists of two muscle layers, an inner circular layer and an outer longitudinal layer (Plate 54, page 604). It differs from the muscularis externa found in the rest of the digestive tract in that the upper one-third is striated muscle, a continuation of the muscle of the pharynx. Striated muscle and smooth muscle bundles are mixed and interwoven in the muscularis externa of the middle third of the esophagus; the muscularis externa of the distal third consists only of smooth muscle, as in the rest of the digestive tract. The mucosa consists of a relatively thick stratified squamous epithelium, a thin layer of lamina propria containing occasional lymphatic nodules, and muscularis mucosae. Mucous glands are present in the submucosa; their ducts, which empty into the lumen of the esophagus, are not evident in this section. External to the submucosa in this part of the esophagus is a thick muscularis externa made up of an inner layer of circularly arranged smooth muscle and an outer layer of longitudinally arranged smooth muscle. After entering the abdominal cavity, the short remainder of the tube is covered by serosa, the visceral peritoneum. Mucosal and submucosal glands of the esophagus secrete mucus to lubricate and protect the luminal wall. The muscle of the esophageal wall is innervated by both autonomic and somatic nervous systems. These glands are scattered along the length of the esophagus but are somewhat more concentrated in the upper half. The excretory duct is composed of stratified squamous epithelium and is usually conspicuous when present in a section because of its dilated appearance. Esophageal cardiac glands are named for their similarity to the cardiac glands of the stomach and are found in the lamina propria of the mucosa. They are present in the terminal part of the esophagus and frequently, although not consistently, in the beginning portion of the esophagus. The striated musculature in the upper part of the esophagus is innervated by somatic motor neurons of the vagus nerve, cranial nerve X (from the nucleus ambiguus). The smooth muscle of the lower part of the esophagus is innervated by visceral motor neurons of the vagus (from the dorsal motor nucleus). These motor neurons synapse with postsynaptic neurons whose cell bodies are located in the wall of the esophagus. Mixing and partial digestion of the food in the stomach by its gastric secretions produce a pulpy fluid mix called chyme. The chyme then passes into the small intestine for further digestion and absorption. The stomach is divided histologically into three regions based on the type of gland that each contains. The mucus produced by the esophageal glands proper is slightly acidic and serves to lubricate the luminal wall. Because the secretion is relatively viscous, transient cysts often occur in the ducts. Those glands near the stomach tend to protect the esophagus from regurgitated gastric contents. Under certain conditions, however, they are not fully effective, and excessive reflux results in pyrosis, a condition more commonly known as heartburn. The cardia surrounds the esophageal orifice; the fundus lies above the level of a horizontal line drawn through the esophageal (cardiac) orifice; the body lies below this line; and the pyloric part is the funnel-shaped region that leads into the pylorus, the distal, narrow sphincteric region between the stomach and duodenum. When the stomach is fully distended, the rugae, composed of the mucosa and underlying submucosa, virtually disappear. The rugae do not alter total surface area; rather, they serve to accommodate expansion and filling of the stomach. At higher magnification, numerous openings can be observed in the mucosal surface.
Purchase hydrea from india
In addition withdrawal symptoms generic hydrea 500 mg with mastercard, it is important to understand that the glomerular filtration barrier is not a passive but an active structure that can remodel itself and modify its own permeability. Although albumin is not a usual constituent, it may sometimes be found in urine, indicating that the size of albumin is close to the effective pore size of the filtration barrier. Despite the ability of the filtration barrier to restrict protein, several grams of protein do pass through the barrier each day. Proliferation of parietal epithelial cells is a typical diagnostic feature in certain types of glomerulonephritis (inflammation of the glomerulus). It is the receptacle for the glomerular ultrafiltrate (primary urine) produced by the filtration apparatus of the renal corpuscle. At the urinary pole of the renal corpuscle, the urinary space is continuous with the lumen of the proximal convoluted tubule. Note that a mesangial cell produces extracellular mesangial matrix, which provides support for the glomerular capillaries. The mesangium is most obvious at the vascular stalk of the glomerulus and at the interstices of adjoining glomerular capillaries. They also endocytose and process a variety of plasma proteins, including immune complexes. Maintaining the structure and function of the glomerular barrier is the primary function of the mesangial cells. In the past, it was suggested that contraction of mesangial cells could increase the intraglomerular blood volume and filtration pressure. Recent studies revealed that mesangial contribution to glomerular filtration rate is minimal, and the mesangial cells may function in regulating glomerular distension in response to increased blood pressure. Proliferation of mesangial cells is a prominent feature in the immunoglobulin A (IgA) nephropathy (Berger disease), membranoproliferative glomerulonephritis, lupus nephritis, and diabetic nephropathy. Embryologically, mesangial and juxtaglomerular cells (discussed in a following paragraph) are derived from smooth muscle cell precursors from the metanephric mesenchyme. Although mesangial cells are clearly phagocytotic, they are unusual in the sense that they are not derived from the usual precursor cells of the mononuclear phagocytotic system, the blood-borne monocytes. Juxtaglomerular Apparatus the juxtaglomerular apparatus includes the macula densa, the juxtaglomerular cells, and the extraglomerular mesangial cells. These antihypertensive drugs do not cause the often-dangerous side effects of the diuretics and -blockers that were previously the most commonly used drugs for control of this condition. Decreased perfusion pressure in the kidney triggers the juxtaglomerular cells to release renin into the blood stream and begin the cascade. Blue arrows indicate stimulatory action on the system; red arrows indicate inhibitory feedback and action of pharmaceutical agents. It typically includes several measurements of physical, biochemical, and microscopic urine characteristics such as pH, specific gravity (indirect measurement of ion concentration), bilirubin, concentration of intermediate compounds derived from the fatty acid metabolism known as ketone bodies, hemoglobin, and concentration of the proteins. The excretion of excessive amounts of protein in the urine (proteinuria or albuminuria) is an important diagnostic sign of renal disease and is a key part of urinalysis. Although excessive excretion of protein almost always indicates renal disease, extreme exercise, such as jogging, or severe dehydration may produce increased proteinuria in individuals without renal disease. Microscopic examination of the urine may reveal presence of red and white blood cells, mineral crystals, and pathogenic agents such as bacteria or fungi. Often these elements are enclosed within the cylindrical structures called urinary casts. The matrix of urinary cast is formed by an 85 kDa protein, uromodulin (Tamm-Horsfall protein), that precipitates in the lumen of distal convoluted tubules and collecting ducts during a disease process. At this site, the wall of the tubule contains cells that are referred to collectively as the macula densa. The nuclei of these cells appear crowded, even to the extent that they appear partially superimposed over one another, thus the name macula densa. In this same region, the smooth muscle cells of the adjacent afferent arteriole (and, sometimes, the efferent arteriole) are modified. They contain secretory granules, and their nuclei are spherical, as opposed to the typical elongate smooth muscle cell nucleus. This system plays an important role in maintaining sodium homeostasis and renal hemodynamics (see Folder 20. The granules of the juxtaglomerular cells contain an aspartyl protease called renin, which is synthesized, stored, and released into the bloodstream from the modified smooth muscle cells. In the blood, renin catalyzes the hydrolysis of a circulating 2-globulin, angiotensinogen, to produce the decapeptide angiotensin I. Then, the juxtaglomerular apparatus functions not only as an endocrine organ that secretes renin but also as a sensor of blood volume and tubular fluid composition. The cells of the macula densa monitor the Na concentration in the tubular fluid and regulate both the glomerular filtration rate and the release of renin from the juxtaglomerular cells. The decreased Na concentration in the distal convoluted tubule is believed to be a stimulus for unique ion-transporting molecules expressed on the apical membrane of macula densa cells. These molecules include Na /K /2Cl cotransporters, Na /H exchangers, and pH- and calcium-regulated K channels. These molecules act in a paracrine manner and signal both the underlying juxtaglomerular cells of the afferent arteriole to secrete renin and the vascular smooth muscle cells to contract. An increase in blood volume sufficient to cause stretching of the juxtaglomerular cells in the afferent arteriole may be the stimulus that closes the feedback loop and stops secretion of renin. Aldosterone, in turn, acts on principal cells of connecting tubules and collecting ducts to increase reabsorption of Na and water, as well as K secretion, thereby raising blood volume and pressure. Thus, the volume of the ultrafiltrate is reduced substantially, and the urine is made hyperosmotic. The long loop of Henle and the connecting tubules and collecting ducts that pass parallel to similarly arranged blood vessels, the vasa recta, serve as the basis for the countercurrent multiplier mechanism that is instrumental in concentrating the urine, thereby making it hyperosmotic. The cuboidal cells of the proximal convoluted tubule have the elaborate surface specializations associated with cells engaged in absorption and fluid transport. The apical surface of the cell shows the closely packed microvilli (Mv) that collectively are recognized as the brush border in the light microscope. Extensive numbers of longitudinally oriented mitochondria (M) are present in the cell within the interdigitating processes. The mitochondria are responsible for the appearance of the basal striations seen in the light microscope, particularly if the extracellular space is enlarged. This higher magnification of the microvilli shows the small endocytotic vesicles that have pinched off from the plasma membrane at the base of the microvilli. In well-fixed histologic preparations, the basal striations and the apical brush border help to distinguish the cells of the proximal convoluted tubule from those of the other tubules. The drawing, at the electron microscopic level, shows the sectioned face on the right and a three-dimensional view of the basolateral surface of a cell with a partial cut face on the left. Here the interdigitating parts of the adjoining cell have been removed to show the basolateral interdigitations. The processes are long in the basal region and create an elaborate extracellular compartment adjacent to the basal lamina. In some locations, the microvilli have been omitted, thereby revealing the convoluted character of the apical cell boundary. Of the 180 L/day of ultrafiltrate entering the nephrons, approximately 120 L/day, or 65% of the ultrafiltrate, is reabsorbed by the proximal convoluted tubule. They are responsible for the reabsorption of Na, which is the major driving force for reabsorption of water in the proximal convoluted tubule. As in the intestinal and gallbladder epithelia, this process is driven by active transport of Na into the lateral intercellular space. Here, the fluid is reabsorbed into the vessels of the peritubular capillary network. The proximal convoluted tubule also reabsorbs nearly all glucose, amino acids, and small polypeptides. The proximal convoluted tubule also recovers approximately 98% of the filtered amino acids. These amino acids are absorbed by several amino acid transporters that either exchange Na, H, and K ions (acidic amino acid transporters) or Na and H ions (basic and neutral amino acid transporters). The brush border in the proximal convoluted tubule resembles that of a striated border in the small intestine in that it possesses many peptidases that degrade large proteins into smaller proteins and polypeptides.
Buy discount hydrea 500 mg line
However medications post mi order hydrea with mastercard, because of initial pseudopodial extensions of plasma membrane that contribute to the formation of phagosome, the actin cytoskeleton must be rearranged in a process that requires depolymerization and repolymerization of the actin filaments. Thus, phagocytosis is referred to as clathrin-independent but actin-dependent endocytosis. In this mechanism, receptors for specific molecules, called cargo receptors, accumulate in well-defined regions of the cell membrane. Cargo receptors recognize and bind to specific molecules that come in contact with the plasma membrane. This drawing shows the steps in the phagocytosis of a large particle, such as a bacterium that has been killed as a result of an immune response. The bacterium is surrounded by antibodies attached to the bacterial surface antigens. Fc receptors on the surface of the plasma membrane of the phagocytotic cells recognize the Fc portion of the antibodies. Depolymerizations and repolymerizations of actin filaments produce temporary projections of the plasma membrane called pseudopodia. By targeted delivery of lysosomal enzymes, a phagosome matures into a lysosome that digests its phagocytosed content. Nonbiologic materials such as inhaled carbon particles, inorganic dusts, and asbestos fibers, as well as cellular debris resulting from inflammation, are internalized without involvement of antibodies and Fc receptors. This diagram shows the steps in receptor-mediated endocytosis, a transport mechanism that allows selected molecules to enter the cell. Cargo receptors recognize and bind specific molecules that come in contact with the plasma membrane. Clathrin interactions then assist the plasma membrane to change shape to form a deep depression, a fully formed coated pit that becomes pinched off from the plasma membrane by the protein complex dynamin as a coated vesicle. Selected cargo proteins and their receptors are thus pulled from the extracellular space into the lumen of a forming coated vesicle. After budding and internalization of the vesicle, the coat proteins are removed and recycled for further use. The uncoated vesicle travels to its destination to fuse with a cytoplasmic organelle. Electron micrograph of the cytoplasmic surface of the plasma membrane of A431 cells prepared by the quick-freeze deep-etch technique. This image shows coated pits and clathrin-coated vesicles in different stages of their formation. Note that the coated pits and clathrin-coated vesicles are formed in areas devoid of actin filaments. The small uniform pinocytotic vesicles do not have a clathrin coat and are located in close proximity to actin filaments. Thus, selected cargo proteins and their receptors are pulled from the extracellular space into the lumen of a forming vesicle. The type of vesicle formed as a result of receptor-mediated endocytosis is referred to as a coated vesicle, and the process itself is known as clathrin-dependent endocytosis. Clathrin-coated vesicles are also involved in the movement of the cargo material from the plasma membrane to early endosomes and from the Golgi apparatus to the early and late endosomes. The next step involves sorting and packaging the secretory product into transport vesicles that are destined to fuse with the plasma membrane in a process known as exocytosis. The membrane that is added to the plasma membrane by exocytosis is recovered into the cytoplasmic compartment by an endocytotic process. Proteins that leave the cell by this process are secreted immediately after their synthesis and exit from the Golgi apparatus, as seen in the secretion of immunoglobulins by plasma cells and of procollagen by fibroblasts. In the regulated secretory pathway, specialized cells, such as endocrine and exocrine cells and neurons, the traveling vesicle. This initial interaction provides recognition of the vesicle and recruits the necessary number of tethering proteins to dock the incoming vesicle. Note that secretory vesicles containing protein ready for secretion fill the apical portion of the cells. This process requires an external signaling mechanism for the cell to discharge the accumulated granules. In this case, a regulatory event (hormonal or neural stimulus) must be activated for secretion to occur, as in the release of secretory vesicles by chief cells of the gastric mucosa and by acinar cells of the pancreas. In the past, secretory vesicles containing inactive precursor (zymogen) were called zymogen granules. In addition to excretory pathways, proteins can be transported between the Golgi apparatus and other organelles along endosomal pathways. These pathways are used for delivery of organelle-specific proteins, such as lysosomal structural proteins, into the appropriate organelles. Blue arrows indicate the constitutive pathway in which proteins leave the cell immediately after their synthesis. In cells using this pathway, almost no secretory product accumulates, and thus few secretory vesicles are present in the cytoplasm. Red arrows indicate the regulated secretory pathway in which protein secretion is regulated by hormonal or neural stimuli. After appropriate stimulation, the secretory vesicles fuse with the plasma membrane and discharge their contents. As discussed previously, newly formed vesicles that bud off from the donor membrane (such as cell membrane or Golgi cisternae) can fuse with a number of possible target membranes within the cell. Shortly after budding and shedding its clathrin coat, a vesicle must be targeted to the appropriate cellular compartment. A targeting mechanism can be likened to a taxi driver in a large city who successfully delivers a passenger to the proper street address. These compartments, called early endosomes, are restricted to a portion of the cytoplasm near the cell membrane where vesicles originating from the cell membrane fuse. However, large numbers of vesicles originating in early endosomes travel to deeper structures in the cytoplasm called late endosomes. Endosomes can be viewed either as stable cytoplasmic organelles or as transient structures formed as the result of endocytosis. This deep-etch electron micrograph shows the structure of an early endosome in Dictyostelium. Early endosomes are located near the plasma membrane and, as in many other sorting compartments, have a typical tubulovesicle structure. The tubular portions contain the majority of integral membrane proteins destined for membrane recycling, whereas the luminal portions collect secretory cargo proteins. The lumen of the endosome is subdivided into multiple compartments, or cisternae, by the invagination of its membrane and undergoes frequent changes in shape. Coated vesicles formed at the plasma membrane fuse only with early endosomes because of their expression of specific surface receptors. In the maturation model, early endosomes are formed de novo from endocytotic vesicles originating from the plasma membrane. Therefore, the composition of the early endosomal membrane changes progressively as some components are recycled between the cell surface and the Golgi apparatus. This maturation process leads to formation of late endosomes and then to lysosomes. Endosomes destined to become lysosomes receive newly synthesized lysosomal enzymes that are targeted via the mannose-6-phosphate (M-6-P) receptor. This pathway provides constant delivery of newly synthesized lysosomal enzymes, or hydrolases. This heavily glycosylated protein then folds in a specific way so that a signal patch is formed and exposed on its surface. This recognition signal is created when specific amino acids are brought into close proximity by the three-dimensional folding of the protein.


