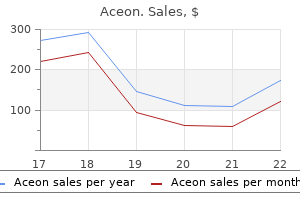
Purchase aceon in india
It is unclear whether airway ciliated cells develop from an undifferentiated precursor or through the further development of a population of secretory cells arrhythmia jaw pain cheap 8mg aceon mastercard. The proximal airways develop basal cells from week 11, and ciliogenesis is complete at birth in humans. Mucous glands develop by 12 weeks and enlarge in the submucosa; secretory activity has been identified in the trachea at 14 weeks. The splanchnopleuric mesenchyme condenses around the epithelium and differentiates into connective tissue cell types and smooth muscle, which differentiates proximal to the tips of the developing airways as they develop, from week 6 onwards. First-trimester human tracheal smooth muscle cells have a fluctuating resting membrane potential that is associated with the spontaneous development of tone and peristalsis-like contractions of the airway of a myogenic nature, probably modified by neurohumoral factors. These contractions are important in moving fluid from distal to proximal within the airways, which is essential for normal lung development and may also stimulate mediator release. Cartilage also develops during this period, and is found in the airways in an adult distribution by 24 weeks. By the end of this period, the airway branching and the pre-acinar vascular patterns are fully mature. Endothelial development is also seen in the pseudoglandular phase when capillary networks form around the developing lung buds. The mesenchyme produces both the endothelium and the smooth muscle cells of the tunica media of the vessels. Vimentin occurs in the cells around developing vessels in the pseudoglandular stage, but is replaced by desmin in the saccular phase. Lung development is described histologically as progressing through embryonic, pseudoglandular, canalicular, saccular and alveolar phases. The investing splanchnopleuric mesenchyme surrounding the lung buds contains a mixed population of cells. Further mesenchymal cells will differentiate into the smooth muscle cells that surround both the respiratory tubes and the blood vessels. In stage 13 embryos, proliferation of the adjacent splanchnopleuric coelomic epithelium (of the primary pleural cavities) is especially evident. The proliferative activity decreases in stage 14, and the mesenchyme becomes arranged in zones around the developing endoderm. Sonic hedgehog is important in lung development; it is tightly regulated and over-expression leads to the formation of excessive lung mesenchyme. The three members of the gli family of transcription factors are implicated in Sonic hedgehog signalling. It forms an extensive capillary network around each lung bud, receiving blood from the developing sixth aortic arch artery and draining it into an anastomosis connected to the dorsal surface of the left atrium in the mediastinal mesenchyme. At 23 weeks, longitudinal sections of the future distal regions show a sawtooth margin, which may indicate the site of further acini. Peripheral growth is accompanied by an increase in the capillary network around the distal airspaces. In many places, the capillaries are in close contact with the respiratory cuboidal epithelium. The primitive cuboidal cells, which hitherto predominated in this part of the lung, differentiate into type 2 epithelial cells (pneumocytes), containing lamellar bodies that form the intracellular storage bodies of surfactant, and type 1 epithelial cells (pneumocytes). Apposition of the capillary networks to the thin pneumocytes (type 1), and reduction of the interstitial tissue of the lung, are prerequisites for future effective gas exchange. By 24 weeks of gestation, the histological characteristics of the airways, including cartilage distribution, are the same as in the adult. Elastin gene expression, possibly modulated through retinoic acid, appears to be of pivotal importance in alveolar budding. Retinoic acid treatment of rats increases alveolarization, and salvages some alveoli in an experimental model, whereas mice with a deletion in the retinoic acid receptor have impaired alveolarization. The pulmonary veins have a similar number of branches but are separated from the airways by the alveoli; they have a different embryological origin, being derived from the mediastinal mesenchyme proximally rather than from the splanchnopleuric mesenchyme around the bifurcating respiratory tree. New vessels are formed by either vasculogenesis (the formation in situ of new blood vessels) or angiogenesis (outsprouting from existing vessels). The earliest pulmonary vessels form in the mesenchyme by vasculogenesis; the capillaries coalesce to form small blood vessels alongside the airways. By 34 days of gestation, blood circulates from the aortic sac via pulmonary arteries into a capillary plexus around the two lung buds and drains to the developing atrium. As each new airway forms in the mesenchyme, a new plexus forms as a halo around it and coalesces with the vessels already alongside the previous airway. In this way, addition of the newly formed tubules to the existing vessels is sustained; the airways act as a template for the development of blood vessels. Initially, this arterial tunica media is derived from the bronchial smooth muscle of the adjacent airways, but as the vessels progressively enlarge, cells derived from the splanchnopleuric mesenchyme differentiate around the wall to form layers of smooth muscle cells. The muscular wall is thick relative to the lumen and this, in part, increases resistance to blood flow, although only 10% of blood flow goes through the pulmonary circulation of the lung during intrauterine life. None the less, this small blood flow is important in normal lung development (see below). At 20 weeks, the structure of the pulmonary vessels is the same as it is in the adult. Vascular innervation follows muscularization; the vasoactive peptides in the nerves are predominantly vasoconstrictor. As the pulmonary veins enlarge, they become separated from the airways by lymphatic vessels lying within connective tissue. At around week 12, the peripheral veins develop a single layer of smooth muscle cells in their walls. This layer is derived only from the splanchnopleuric mesenchyme and not from bronchial smooth muscle (as occurs in the arteries). The lumen of each vein is relatively large and the wall is relatively thin at all levels. Arteries and veins continue to develop in the canalicular phase, probably by angiogenesis; dividing cells are seen in the peripheral capillaries. The epithelium of the most peripheral conducting airways flattens when the developing capillaries come to lie immediately subjacent to it during this phase. Two or three bronchial arteries grow from the descending aorta from week 8 onwards, entering the lung at the hilum. These arteries extend down the intrapulmonary airways, eventually forming subepithelial and adventitial plexuses, which, by birth, have reached the distal portions of the bronchioli. Saccular/alveolar phase (28 weeks to term: first appearance of alveoli in humans) Thin-walled terminal saccules are apparent at the saccular stage and will become alveolar ducts as development proceeds. The expansion of the prospective respiratory airspaces that occurs during this period is accompanied by a further decrease in the amount of interstitial tissue, and the capillary networks become ever more closely opposed to the pneumocyte epithelium. As a crest protrudes into a saccule, part of the capillary network becomes drawn into it. After the later expansion of the saccules on each side of the crest, a double capillary layer becomes annexed between what are now alveolar walls. During the saccular stage, elastin is deposited beneath the epithelium (an important step for future alveolar formation), and surfactant production from type 2 pneumocytes (essential for the survival of a preterm neonate) matures. Alveolar phase Exactly when the saccular structure of the lung can be termed alveolar is not yet clear: estimates range from 28 to 32 weeks. The distal airspaces expand during late gestation and continue to do so after birth. This process is accompanied by fusion of adjacent capillary nets, so that, shortly after birth, there is an extensive double capillary net. The alveolar stage is now considered to continue beyond infancy and to last throughout childhood (see below). The formation of millions of alveoli is accomplished by a complex process of folding and division. Existing walls of distal airspaces become thinner as pneumocyte epithelial cells flatten, and some cells undergo apoptosis. Ridges subsequently grow out from the sides of the saccule walls, forming primitive alveoli.
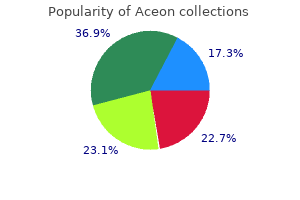
Generic aceon 2 mg with amex
At the lower border of subscapularis hypertension 6 year old buy aceon 8 mg mastercard, it curves back inferior to the humeroscapular articular capsule and, with the posterior circumflex humeral vessels, traverses a quadrangular space bounded above by subscapularis (anteriorly) and teres minor (posteriorly), below by teres major, medially by the long head of triceps, and laterally by the surgical neck of the humerus. The anterior branch curves round the neck of the humerus with the posterior cir cumflex humeral vessels, deep to deltoid, to reach the anterior part of the muscle. At the level of the anterior border of the acromion, the nerve often consists of two or more thin filaments that supply the anterior deltoid; it gives off a few small cutaneous branches that pierce deltoid and ramify in the skin over its lower part. The posterior branch courses medially and posteriorly along the attachment of the lateral head of triceps, inferior to the glenoid rim. It gives off the nerve to teres minor and the upper lateral cutaneous nerve of the arm at the lateral edge of the attachment of the long head of triceps. The posterior branch frequently supplies the posterior aspect of deltoid, usually via a separate branch from the main stem, but occasionally from the superior lateral cutane ous nerve of the arm. However, the posterior part of deltoid has a more consistent supply from the anterior branch of the axillary nerve, which should be remembered when performing a posterior deltoidsplitting approach to the shoulder. The upper lateral cutaneous nerve of the arm pierces the deep fascia at the medial border of the posterior aspect of deltoid, and supplies the skin over the lower part of deltoid and the upper part of the long head of triceps. The posterior branch is intimately related to the inferior aspects of the glenoid and shoulder joint capsule. It runs laterally, deep to trapezius and omohy oid, and enters the supraspinous fossa through the suprascapular notch inferior to the superior transverse scapular ligament. It runs deep to supraspinatus, supplies it, and curves round the lateral border of the spine of the scapula with the suprascapular artery to reach the infrasp inous fossa, where it gives two branches to infraspinatus and articular rami to the shoulder and acromioclavicular joints. When present, it pierces deltoid close to the tip of the acromion and supplies the skin of the proximal third of the arm within the territory of the axillary nerve. Lateral pectoral nerve the lateral pectoral nerve (C5, 6) is larger than the medial, and may arise either from the anterior divisions of the upper and middle trunks, or by a single branch from the lateral cord, just above or deep to the clavicle. It crosses anterior to the axillary artery and vein, pierces the clavipectoral fascia and supplies the deep surface of pectoralis major. It sends a branch to the medial pectoral nerve, forming a loop in front of the first part of the axillary artery to supply some fibres to pectoralis minor. The delay before diagnosis is inexplicable despite the reliability of precise but elementary clinical examination. Matters are not helped by the persistence with which incorrect and discredited ideas about what muscles do continue to be taught. Many nerve injuries in this region are painful and, too often, a patient is passed to a pain clinic without a diagnosis. This range of movement occurs at the shoulder joints, mainly the glenohumeral joint, with some involve ment of the acromioclavicular and sternoclavicular joints. It is important to remember that full elevation is possible the integrity of the accessory nerve is fundamental to thoracoscapular function and essential for scapulohumeral rhythm. The nerve is usually divided at the apex of the posterior triangle, usually by surgeons, and is a crippling injury. Typically, there is immediate pain, then dropping of the shoulder and a remarkable loss of function. The pain is characteristic: often severe, deepseated, dull, constant and boring in nature, and felt around the scapula and the posterior aspect of the neck and shoulder. It is very hard to examine muscles in a patient with a fracture disloca tion of the shoulder. Consider, for example, a patient after successful reduction of a dislocated shoulder, whose arm is supported in a sling. All too often, rupture of the axillary nerve is not recognized until deltoid is atrophied, long past the ideal time for repair. Deltoid provides power and stamina, and abduction is initiated and maintained by the suprascapular nerve, acting through supra and infraspinatus, their tendons and attachments. Disruption of any component of this abduc tor mechanism interferes with or precludes abduction. The range of abduction was full or nearly so, and a system of training enabled most patients to return to full military duties. He wrote: It must be stressed that these movements providing full abduction and elevation are not trick movements, in the sense usually associated with this word; all the muscles involved normally help to abduct the shoulder. The scapulohumeral rhythm is quite normal and in the later stages of re-education the patient does not even need to rotate the humerus externally to initiate movement. Scapular winging, without prominence of the lower fibres of trapezius, in a case where there is some early recovery into the upper fibres after repair of the accessory nerve. It is fortunate that repair of the nerve is usually successful, even when repair is performed as late as 4 years after injury, with early relief of pain long before there is re innervation of muscle (Camp and Birch 2011). The nerve appears to be relatively immune to the harmful effects of delayed repair. The suprascapular nerve is essential for abduction and lateral rotation at the glenohumeral joint. Pain and loss of function are usual and are only slightly less than those seen after accessory nerve palsy. This action is increased by the weight of the limb, which contributes to the winging and is most notable in protraction. Although a good range of eleva tion is usually maintained by an intact abductor mechanism, strength and stamina are greatly reduced. Patients with an intact supra scapular nerve and an intact rotator cuff are able to initiate abduction and many can fully elevate the arm, or nearly so. One useful test is to palpate the posterior third of deltoid with the fingers and ask the patient to move the elbow gently backwards. This is easily distinguishable from the winging provoked by accessory palsy by the position of the scapula, which is drawn upwards and towards the spine by the unopposed action of trapezius, levator scapulae and the rhomboids. A, the range of elevation at the right shoulder in a 74-year-old man, in whom there was clear evidence of recovery for both of the nerves. The weight of the man and the force of the current were such that he felt the muscles tearing in his right shoulder; then he felt the head of the humerus pulling out from the socket and his arm went dead. Subscapular nerves to subscapularis and teres major Thoracodorsal nerve Latissimus dorsi Medial cutaneous nerve of forearm Medial cutaneous nerve of arm 48. It pierces coracobrachialis a variable distance below the tip of the coracoid process and then descends laterally between biceps brachii and brachialis to the lateral side of the arm. The first branch, to coraco brachialis, may pass into the muscle no more than 2 cm distal to the tip of the coracoid; fibres from C7 may branch directly from the lateral cord. Branches to biceps brachii and brachialis leave after the musculo cutaneous nerve has pierced coracobrachialis. There are usually two branches to brachialis; the lower branch also supplies the anterior aspect of the elbow joint. The nerve now inclines lateral and deep, into the plane between brachialis posteriorly and biceps anteri orly, where it supplies a small branch to the humerus that enters the shaft with the nutrient artery. Below the branches to brachialis, the musculocutaneous nerve continues as the lateral cutaneous nerve of the forearm. It may run behind coracobrachialis or adhere for some distance to the median nerve and pass behind biceps brachii. Some fibres of the median nerve may run in the musculocutaneous nerve, leaving it to join their proper trunk; less frequently, the reverse occurs, and the median nerve sends a branch to the musculocutaneous nerve. Occasionally, it supplies prona tor teres and may replace radial branches to the dorsal surface of the thumb. It lies initially between the axillary artery and vein, and gives off a ramus that pierces the deep fascia to supply the skin over the biceps, almost to the elbow. The nerve descends medial to the bra chial artery, pierces the deep fascia with the basilic vein midway in the arm, and divides into anterior and posterior branches. Some fibres from C7 often leave the lateral root in the lower part of the axilla; they pass distomedially posterior to the medial root, and usually ante rior to the axillary artery, to join the ulnar nerve. Clinically, they are believed to be mainly motor and to supply flexor carpi ulnaris.
Order aceon 2 mg on-line
Imaging in diagnostic or interventional procedures may produce still or motion (cine) images arteria zigomatica discount 4mg aceon. Angiograms of the heart may be performed to visualize the size and contractility of the chambers and the anatomy of the coronary vessels. The thorax may also be studied to evaluate the pulmonary arteries and veins for vascular malformations, blood clots and possible origins of haemoptysis. In the investigation of atherosclerotic disease, vascular malformations or tumoral vascularization, the neck is often imaged in order to visualize the vessels that supply the brain in their entirety, from the points at which they arise from the aortic arch to their termination as cerebral vessels. Renal artery imaging may elucidate the cause of hypertension in selected patients, and imaging of the mesenteric vessels may identify the origin of gastrointestinal bleeding or mesenteric angina. In addition to angiograms and venograms, the field of interventional radiology also includes such procedures as coil embolization of aneurysms and vascular malformations; balloon angioplasty and stent placement; chemoembolization directly into tumours; drainage catheter insertion; embolization. All of the tissues contained within the pixel attenuate the X-ray projections and result in a mean attenuation value for the pixel. This value is compared with the attenuation value of water and is displayed on a scale (the Hounsfield scale). The fast scan times allow dynamic imaging of arteries and veins at different times after the injection of intravenous contrast agents. This orthogonal imaging greatly improves the understanding of the threedimensional aspects of radiological anatomy and now forms part of the standard practice of assessing disease. Studies of the chest, abdomen and pelvis, and those of the brain with complex histories, usually require intravenous contrast medium that contains iodine because this defines vascular relationships and differentiates normal and pathological soft tissues more effectively. This procedure is much less frequently performed with the latest generation of scanners, which exquisitely differentiate various enhancing layers within the bowel wall. Occasionally, direct insertion of rectal contrast to show the distal large bowel may be required. Generally, all studies are performed with the patient supine and images are obtained in the transverse or axial plane. Occasionally, direct coronal images are obtained in the investigation of cranial and maxillofacial abnormalities; in these cases, the patient lies prone with the neck extended and the gantry appropriately angled, but this technique has largely been superseded by the orthogonal imaging described above. Foucher J, Chanteloup E, Vergniol J et al 2006 Diagnosis of cirrhosis by transient elastography (FibroScan): a prospective study. Where endoscopic staging showed no evidence of locally advanced disease, the patient underwent surgical staging (n = 65). Sensitivity was significantly greater for combined surgical/endoscopic staging (94% versus 79%, P = 0. Prior to this, surgery had been the only way to obtain tissue from mediastinal masses or lymph nodes. Biopsies could be taken by inserting standard biopsy forceps through a sheath left in place to mark the correct position once the ultrasound catheter was removed. In 2003, the first report of a prototype bronchoscope, incorporating a curved linear array electronic transducer at the end of the bronchoscope, was published (Krasnik et al 2003). When the trans ducer was closely applied to the bronchial wall, ultrasound scanning of mediastinal structures (to a depth of 5 cm) was possible and a 22gauge needle inserted through the biopsy channel and into the mass in ques tion was visible in real time. This meant that smaller lesions could be biopsied more accurately, with reduced potential for the wrong area to be sampled inadvertently. These lymph nodes or masses are then found during bronchoscopy using the ultrasound scanning probe. The infla tion of a small waterfilled balloon around the scanning probe can improve contact with the bronchial wall, and thus image quality, although in most cases it is not needed. A needle is inserted through the working channel of the bronchoscope and suction applied, via a syringe, at the proximal end of the needle. Under direct vision, the needle is passed several times through the lesion in order to obtain a cytological specimen. The specimen is either smeared on to slides and airdried, or placed into pots containing liquid preservative for laboratory processing (Medford et al 2010). The power Doppler facility enables safer identification of vessels to avoid puncture of these structures. Surgical sampling of the mediastinum, whilst remaining the gold standard, has a number of limitations, including invasiveness and cost. In 2009, a new lymph node map was published by the International Association for the Study of Lung Cancer (Rusch et al 2009). The future In this era of increasingly personalized medicine, it is important for any sampling technique to provide sufficient tissue for molecular analysis. Ojha S, Ie S, Boyd M et al 2014 Vertebral body tumor biopsy: an expanded role of endobronchial ultrasoundguided transbronchial needle aspira tion. Ortiz R, Hayes M, Arias S et al 2014 Pneumomediastinum and pneumoperi cardium after endobronchial ultrasoundguided transbronchial needle aspiration. Sun J, Teng J, Yang H et al 2013 Endobronchial ultrasoundguided trans bronchial needle aspiration in diagnosing intrathoracic tuberculosis. Yarmus L, Akulian J, Gilbert C et al 2013 Optimizing endobronchial ultra sound for molecular analysis. Yasufuku K, Pierre A, Darling G et al 2011 A prospective controlled trial of endobronchial ultrasoundguided transbronchial needle aspiration compared with mediastinoscopy for mediastinal lymph node staging of lung cancer. Together, they provide multiple vital functions, including: housing and protection of the digestive and urinary tracts and of the internal reproductive organs; a conduit for neurovascular communication between the thorax and lower limb; support and attachment for the external genitalia; access to and from the internal reproductive organs and urinary tract; assistance with physiological functions such as respiration, defecation and micturition; support for the vertebral column in weight-bearing, maintenance of posture and movement; and, in women, the ability to support human gestation. In addition, the upper abdominal cavity gains protection from the lower six ribs and their cartilages, even though these structures are technically part of the thoracic wall. The anterolateral muscles, in particular, provide assistance with rotation of the thorax in relation to the pelvis (or vice versa if the thorax is fixed). The abdominal cavity is somewhat kidney-shaped in horizontal cross-section due to the posterior indentation of the vertebral column. Consequently, there are two distinct paravertebral gutters on either side of the spine. The lordosis of the lumbar spine combined with the backward angulation of the sacrum gives each paravertebral gutter a parabola shape in sagittal section. Sympatheticinnervation the cell bodies of neurones of the sympathetic supply of the abdomen and pelvis lie in the intermediolateral grey matter of the first to the twelfth thoracic and the first two lumbar spinal segments. Myelinated axons from these neurones travel in the ventral ramus of the spinal nerve of the same segmental level, leaving it via a white ramus communicans to enter a thoracic or lumbar paravertebral sympathetic ganglion. Visceral branches may exit at the same level or ascend or descend several levels in the sympathetic chain before exiting; they leave the ganglia without synapsing and pass medially, giving rise to the paired greater, lesser and least splanchnic nerves, and the lumbar and sacral splanchnic nerves. Axons destined to supply somatic structures synapse in the sympathetic ganglion of the same level, and postganglionic, unmyelinated axons leave the ganglion as one or more grey rami communicantes to enter the spinal nerve of the same segmental level. Thoracoabdominalinterface the diaphragm constitutes the interface between the thoracic and abdominal cavities (Ch. Three principal pathways exist between the two cavities across the diaphragm: the caval opening in the central tendon transmits the inferior vena cava and right phrenic nerve; the oesophageal hiatus, encircled by the right crus of the diaphragm, transmits the oesophagus, vagal trunks and vessels; and the aortic hiatus, posterior to the median arcuate ligament of the diaphragm, transmits the aorta, thoracic duct and, usually, the azygos vein. The hemiazygos vein usually enters the thorax through the left crus of the diaphragm. Other lymphatics from the abdomen drain to the thorax alongside the inferior vena cava and via small vessels passing through and around the diaphragm. Thoracic splanchnic nerves reach the abdomen through the diaphragmatic crura and behind the medial arcuate ligaments, and the left phrenic nerve pierces the muscle of the left hemidiaphragm. The subcostal vessels pass into the abdomen beneath the lateral arcuate ligaments of the diaphragm. Anteriorly, the superior epigastric vessels pass between the costal and xiphoid attachments of the diaphragm. Neurovascular structures also cross between the thorax and abdomen within the subcutaneous tissues. Greater splanchnic nerve On each side, the greater splanchnic nerve is derived from the medial, visceral branches of the fifth to ninth thoracic sympathetic ganglia.

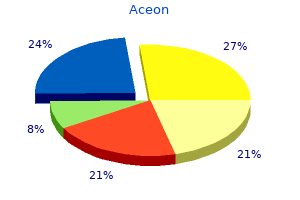
Generic 8 mg aceon with visa
As it proceeds distally arrhythmia when sleeping buy discount aceon 4 mg line, abductor pollicis longus becomes more superficial in the forearm, where it is visible as an oblique elevation. It runs between abductor 854 muscles of the forearm pollicis longus and extensor pollicis longus. The tendon is attached to the base of the proximal phalanx of the thumb and, commonly, has an additional attachment to the base of the distal phalanx, usually through a fasciculus that joins the tendon of extensor pollicis longus. Extensor pollicis brevis may be absent or fused completely with abductor pollicis longus. Humerus Brachioradialis Relations Extensor pollicis brevis is ulnar (medial) to , and closely connected with, abductor pollicis longus. They cover the distal part of brachioradialis, and pass through the most lateral compartment of the extensor retinaculum in a single synovial sheath, sharing a groove in the distal radius. Ultimately, they cross, superficial to the radial styloid process and radial artery, to reach the dorsolateral base of the proximal phalanx of the thumb. Biceps brachii Median nerve Vascular supply Extensor pollicis brevis is supplied from branches from the posterior interosseous artery, together with perforating branches from the anterior interosseous artery. Posterior interosseous nerve Biceps brachii, tendon Supinator Pronator teres Palmaris longus Flexor carpi radialis Innervation Extensor pollicis brevis is innervated by the posterior interosseous nerve, C7 and 8. Actions Extensor pollicis brevis extends the proximal phalanx and metacarpal of the thumb. Testing the tendon of extensor pollicis brevis can be felt at the radial border of the anatomical snuff-box, lying medial to the tendon of abductor pollicis longus, when the metacarpophalangeal joint of the thumb is extended against resistance. It arises from the posterior surface of the ulna distal to extensor pollicis longus and the adjacent interosseous membrane. Opposite the head of the second metacarpal, it joins the ulnar side of the tendon of extensor digitorum that serves the index finger. Extensor indicis occasionally sends accessory slips to the extensor tendons of other digits. Its tendon may be interrupted on the dorsum of the hand by an additional muscle belly (extensor indicis brevis manus). Pronator quadratus Relations the posterior interosseous artery passes over the muscle belly of extensor indicis. On the dorsum of the hand, the tendon of extensor indicis lies on the ulnar aspect of the tendon of extensor digitorum to the index finger. Vascular supply Extensor indicis is supplied on its superficial surface by branches from the posterior interosseous artery, and on its deep surface by perforating branches from the anterior interosseous artery. Innervation Extensor indicis is innervated by the posterior interosseous nerve, C7 and 8. Actions Extensor indicis extends the index finger independently of the other digits. Relations Supinator emerges under the lower border of anconeus and is related to abductor pollicis longus at its inferior medial border. The posterior interosseous nerve enters the forearm by passing between its superficial and deep heads. Testing Extensor indicis is tested by extending the index finger while holding the remaining fingers flexed at the metacarpophalangeal joints in order to eliminate the effects of any simultaneous contraction of extensor digitorum. Vascular supply the superficial part of supinator is supplied by branches from the radial recurrent artery. The deep part of supinator is supplied by branches from the posterior interosseous artery and the posterior interosseous recurrent artery. Supinator Attachments Supinator is a wide muscle that surrounds the proximal third of the radius. Supinator is attached distally to the lateral surface of the proximal third of the radius, down to the insertion of pronator teres. Supinator is subject to frequent Innervation Supinator is innervated by the posterior interosseous nerve, C6 and 7. It acts alone in slow, unopposed supination, and together with biceps brachii in fast or forceful supination. An object, which potentially may be heavy, is often picked up with the forearm initially pronated. The more powerful supinator lifts the object against gravity; rotation is often combined with increasing elbow flexion in order to bring the object towards the eyes. The latter separates the artery from the anterior structures, namely: the skin, superficial fascia and the median cubital vein. It passes deep to brachioradialis and gives off the radial recurrent artery before continuing into the forearm. It descends along the lateral side of the forearm, accompanied by paired venae comitantes, from the medial side of the neck of the radius to the wrist. Proximally, it is overlapped anteriorly by the belly of brachioradialis, but elsewhere in its course it is covered only by the skin and superficial and deep fasciae. Its posterior relations in the forearm are, successively, the tendon of biceps, supinator, the distal attachment of pronator teres, the radial head of flexor digitorum superficialis, flexor pollicis longus, pronator quadratus and the lower end of the radius (where its pulsation is Elbow and forearm A compartment syndrome exists where increased pressure in an osseofascial compartment leads to decreased perfusion of the contents of the compartment. In the forearm, this commonly occurs as a result of trauma, external pressure, burns, arterial injury or post-ischaemic swelling. The resultant increase in interstitial pressure causes vascular occlusion and myoneural ischaemia. The patient presents with pain that is disproportionate to the severity of the injury and which is provoked or markedly increased by passive extension of the fingers. If a compartment syndrome is suspected, compartment pressure measurements may be indicated, but, in unequivocal cases, the definitive treatment is emergency fasciotomies of the forearm. The absolute intracompartmental pressure threshold for decompression is the subject of debate: an absolute value of 30 mmHg is generally accepted, although some authorities prefer a figure that comes within 30 mmHg of diastolic pressure. Incisions to decompress the flexor compartment (Ronel et al 2004) run from the medial epicondyle and curve down the anterior aspect of the forearm, ending on the radial side of flexor carpi ulnaris; from here, they may be extended distally to decompress the carpal tunnel, if necessary. The bicipital aponeurosis (lacertus fibrosus) is routinely released to decompress the median nerve. After retraction of the superficial flexors, the fascia over the deep muscles of the forearm may be incised over its full length. Both median and ulnar nerves should be inspected to ensure that their decompression is complete. The mobile wad and dorsal compartment are decompressed through a straight longitudinal incision that starts just distal to the lateral epicondyle; this is deepened through the plane between extensor digitorum and extensor carpi radialis brevis, and ends over the midline of the wrist. In both cases, the wounds are left open and dressed, and only closed when circumstances permit. Pronator teres is medial to the proximal part of the artery, and the tendon of flexor carpi radialis is medial to the distal portion. The superficial radial nerve lies lateral to the middle third of the radial artery; multiple branches from the artery supply the nerve throughout its length. The radial artery may occasionally arise from a continuation of a superficial brachial artery, or as a high proximal division of an otherwise normal brachial artery. It passes between the superficial radial nerve and the posterior interosseous nerve before ascending beneath brachioradialis, anterior to supinator and brachialis. It supplies these muscles and then anastomoses with the radial collateral branch of the profunda brachii artery. The radial recurrent artery also gives branches to extensor carpi radialis longus, extensor digitorum, extensor digiti minimi and extensor carpi ulnaris. Ulnar collateral ligament Extensor carpi ulnaris (cut tendon) Anconeus (cut tendon) Anular ligament Interosseous recurrent artery Supinator Anconeus Posterior interosseous nerve Extensor carpi ulnaris Flexor carpi ulnaris Flexor digitorum superficialis Posterior ulnar recurrent artery Flexor digitorum profundus Muscular branches Muscular branches are distributed to the muscles on the radial side of the forearm. Direct branches from the radial artery supply the radial insertion of pronator teres, the anterolateral aspect of flexor carpi radialis, the lateral surface of flexor digitorum superficialis, the lateral half of flexor pollicis longus, and the distal sections of extensor carpi radialis longus and extensor carpi radialis brevis. It is larger than the radial artery and is accompanied throughout its length by venae comitantes. In the forearm, the ulnar artery initially lies on brachialis before passing deep to pronator teres, flexor carpi radialis, palmaris longus and flexor digitorum superficialis. It then lies on flexor digitorum profundus, between flexor carpi ulnaris and flexor digitorum superficialis, and is covered by the skin and superficial and deep fasciae. The artery crosses the flexor retinaculum, lateral to the ulnar nerve and pisiform bone, to enter the hand.
Discount aceon 8 mg with mastercard
As the muscle curves round teres major blood pressure what is high discount 4 mg aceon, the fasciculi rotate around each other, so that fibres that originate lowest at the midline insert highest on the humerus, and fibres that originate highest at the midline insert lowest on the humerus. The tendons of latissimus dorsi and teres major are united at their lower borders but separated by a bursa near their humeral attachments. When the arm is adducted against resistance, this fold is accentu ated, and the whole inferolateral border of latissimus dorsi can be traced to its attachment to the iliac crest. The lower, lateral margin of latissimus dorsi is usually separated from the posterior border of external oblique by the lumbar triangle. The base of this small triangle is the iliac crest and its floor the internal oblique; it should not be confused with the triangle of auscultation, medial to the scapula, which is bounded above by trapezius, below by latissimus dorsi and laterally by the medial border of the scapula. If the scapulae are drawn forwards, by folding the arms across the chest, and the trunk is bent forwards, parts of the sixth and seventh ribs and the interspace between them (overlying the apex of the lower pulmonary lobe) become subcutaneous. Variants Latissimus dorsi commonly receives some additional fibres from the scapula as it crosses the inferior scapular angle. A muscular Latissimus dorsi Vascular supply Latissimus dorsi is supplied by a single dominant vascular pedicle, the thoracodorsal artery, itself a continuation of the subscapular artery. The artery gives off up to three large branches to serratus anterior before dividing at, or even before, the neurovascular hilum for latissimus dorsi. This usually yields a small recurrent branch that returns to supply the proximal part of the muscle but, in some cases, it provides a third major branch that supplies the distal part of the muscle. Occasionally, the lateral branch gives off a further collateral to serratus anterior. Musculocutaneous perforators arising from these vessels supply the overlying skin. In addition to the dominant vascular pedicle, latissimus dorsi is supplied inferiorly by several smaller, secondary, segmental vascular pedicles. These are dorsal perforating arteries derived from the ninth, tenth and eleventh posterior intercostal arteries and the first, second and third lumbar arteries. They all enter the muscle on its deep surface and anastomose within the muscle with branches of the thoracodorsal artery. Innervation Latissimus dorsi is supplied by the thoracodorsal nerve, from the posterior cord of the brachial plexus, C6, 7 and 8 (predomi nantly C7). The neural and vascular branches travel together and so the pattern of branching of the nerve follows that of the artery closely. Actions Latissimus dorsi is active in adduction, extension and, espe cially, in medial rotation of the humerus. Humeral adduction and extension are most powerful when the initial position of the arm is one of partial abduction or flexion, or a combination of the two. With the sternocostal part of pectoralis major and teres major, it adducts the raised arm against resistance. When the arms are raised above the head, as in climbing, it pulls the trunk upwards and forwards. It takes part in all violent expiratory efforts, such as coughing or sneezing; this is readily confirmed by palpation. Electromyography suggests that latis simus dorsi aids deep inspiration, but it is also active towards the end of forcible expiration. When the arm is elevated, the stretched fibres of latissimus dorsi press on the inferior scapular angle (to which it may also be attached), keeping it in contact with the chest wall. Despite this range of actions, surgical transposition of the muscle does not appear to produce any serious restriction of normal activity. Since teres minor, parts of infraspinatus and most of subscapularis do not have tendons, this use of the term excludes impor tant parts of the rotator cuff apparatus. It is also maintained that the inferior capsule of the glenohumeral joint is exposed as a weak part of the capsule, and so predisposed to 822 muscles injury (dislocation). Teres major is a part of the cen tralizing function of the rotator cuff, and should be considered along with the conventional definition of the rotator cuff. The muscles of the rotator cuff are attached to their tendons in a similar manner: the fibrous architecture of the tendons provides a framework for the action of the rotator cuff so that it always generates a centralizing force on the glenoid (Gagey et al 1993). The muscles of the rotator cuff combine in three groups (subscapu laris and teres major, supraspinatus and infraspinatus, and teres minor) by virtue of their innervation. Subscapularis and teres major are innervated through the subscapular nerves (C5, 6 and 7, posterior divi sion, posterior cord); supraspinatus and infraspinatus by the supras capular nerve (C5, 6, upper trunk); and teres minor by the axillary nerve (C5, 6, posterior division, posterior cord). The common origin of their innervation suggests a common final action, namely: the gen eration of the centralizing compression force. In functional terms, supraspinatus and infraspinatus can be considered as one muscle on the grounds that they have the same innervation and the same vascular supply; their attachment to the scapula is a continuum broken only by the spine of scapula for the attachment of trapezius; and their actions are complementary. It arises by mus cular fibres from the medial twothirds of the fossa, by tendinous fibres from ridges on its surface and from the deep surface of the infraspinous fascia, which separates it from teres major and minor. Its fibres converge to a tendon that glides under the lateral border of the spine of the scapula, and then passes across the posterior aspect of the capsule of the shoulder joint to be attached to the middle facet on the greater tubercle of the humerus. The tendon fuses with that of supraspinatus immediately lateral to the thick lateral margin of the spine of the scapula; the tendon fibres decussate so that fibres of infra spinatus can be traced to the facet for supraspinatus, and vice versa (Mochizuki et al 2008). The tendon is sometimes separated from the capsule by a bursa, which may communicate with the joint cavity. The tendon is flattened, but is thickened in several places, reinforcing its posterior aspect as it attaches to the middle facet. From this region, fibres passing orthogonally within the deeper substance of the tendon of infraspinatus pass forwards into supraspinatus, then pass across the rotator interval to subscapularis. The muscular part of infraspinatus is attached directly to the middle facet on the greater tubercle and is sometimes fused with teres minor laterally, although their tendons are usually separable closer to the joint capsule. Vascular supply Infraspinatus is supplied by the suprascapular and circumflex scapular arteries. The fibres converge, under the acromion, into a thick tendon that passes obliquely through the muscle, crosses above the humeral head, and is attached to the highest facet of the greater tubercle of the humerus. The tendon has a posterior, flatter, component closer to the facet of attach ment. The anterior, obliquely directed, fibres of supraspinatus are inserted into the anterior aspect of the thick tendon, while the more parallel posterior fibres are inserted into both the thick tendon and the flatter component. The thick tendon is contiguous with the transverse humeral and coracohumeral ligaments; it forms an anterior pillar to the posterosuperior part of the rotator cuff. The deep surface of the tendon blends into the articular capsule over its distal 1 cm or so. Fibrocartilage has been described at the tendinous insertion, as in other tendons attached to epiphysial bone. Together with supraspinatus, subscapularis and teres minor, it helps to stabilize the head of the humerus in the glenoid fossa during shoulder movements. The upper fibres end in a short tendon (which may be absent), attached to the lowest facet on the greater tubercle of the humerus. The lower fibres are attached directly into the humerus distal to this facet and above the origin of the lateral head of triceps. The tendon passes across, and blends with, the lower posterior surface of the capsule of the shoulder joint. Vascular supply Supraspinatus is supplied by the suprascapular and dorsal scapular arteries. Actions the conventional view is that supraspinatus initiates abduc tion of the shoulder and assists deltoid in abduction thereafter. However, there is evidence that both supraspinatus and deltoid are involved throughout the range of abduction, including initiation of the movement.
Order line aceon
There are four principal pathways between the pelvis and lower limb: the interval beneath the inguinal ligament anterior to the superior pubic ramus and ilium arrhythmia icd 9 order 2 mg aceon overnight delivery, which transmits the femoral neurovascular structures and lymphatics; the greater and lesser sciatic foramina, which transmit the gluteal vessels and nerves, sciatic nerve, and internal pudendal vessels and pudendal nerve; and the obturator foramen, which transmits the obturator nerve, vessels and lymphatics. Autonomic nerves travel with the arterial supply to the lower limb and with the branches of the sacral plexus. Neurovascular structures also cross between the lower limb and pelvis within the subcutaneous tissues. On each side, the lesser splanchnic nerve is derived from the medial, visceral branches of the tenth and eleventh thoracic ganglia (or ninth and tenth). It enters the abdomen running through the lowermost fibres of the ipsilateral crus of the diaphragm or under the medial arcuate ligament, and then lies on the crus as it runs anteroinferiorly. The trunk of the nerve joins the aorticorenal ganglion and may give branches to the lateral aspect of the coeliac ganglion. It occasionally joins the greater and least splanchnic nerves as a single splanchnic nerve. It enters the abdomen medial to the sympathetic chain under the medial arcuate ligament of the diaphragm and runs inferiorly to join the renal plexus. The trunk of the nerve enters the aorticorenal ganglion and may give branches to the lateral aspect of the coeliac ganglion. It is sometimes part of the lesser splanchnic nerve, when it forms a twig that enters the renal plexus just below the aorticorenal ganglion. The thoracic splanchnic nerves are subject to considerable individual variation in their origin and distribution (Loukas et al 2010). For example, the greater splanchnic nerve may receive a contribution from the fourth or tenth thoracic sympathetic ganglia or originate only from the sixth to ninth ganglia. Superiorly, it is continuous with the thoracic sympathetic trunk posterior to the medial arcuate ligament. Inferiorly, it passes posterior to the common iliac vessels and is continuous with the sacral sympathetic trunk. It is anterior to most of the lumbar vessels but may pass behind some lumbar veins. On each side, the first, second and, sometimes, the third lumbar ventral spinal rami are connected to the lumbar sympathetic trunk by white rami communicantes. All lumbar ventral rami are joined near their origins by long, slender, grey rami communicantes from the four lumbar sympathetic ganglia. The autonomic supply to the abdominal and pelvic viscera is via the abdominopelvic part of the sympathetic chain and the greater, lesser and least splanchnic nerves (sympathetic), and the vagus and pelvic splanchnic nerves (parasympathetic). The details of the terminations of these fibres are given in the description of the microstructure of the gut wall. As a general rule, sympathetic neurones from the abdominopelvic autonomic plexuses inhibit visceral smooth muscle motility and glandular secretions, induce sphincter contraction and cause vasoconstric- 8 Somatic and vascular branches Sympathetic nerve branches accompany the lumbar arteries round the sides of the vertebral bodies, medial to the fibrous arches to which psoas major is attached, to provide sympathetic innervation to the lumbar somatomes. The first lumbar splanchnic nerve, from the first ganglion, gives branches to the coeliac, renal and inferior mesenteric plexuses. The second nerve joins the inferior part of the intermesenteric or inferior mesenteric plexus. The third nerve arises from the third or fourth ganglion and joins the superior hypogastric plexus. The fourth lumbar splanchnic nerve from the lowest ganglion passes anterior to the common iliac vessels to join the lower part of the superior hypogastric plexus, or the hypogastric nerves. It is important to note that lumbar splanchnic sympathetic nerves contribute to the superior and inferior hypogastric plexuses and, therefore, contribute to the innervation of the bladder neck, ductus deferens and prostate, among other structures. Other postganglionic sympathetic nerves to vessels and skin travel with somatic nerves. Thus, the femoral nerve carries vasoconstrictor sympathetic nerves to the femoral artery and its branches in the thigh, as well as sympathetic fibres in its cutaneous branches. Postganglionic fibres travelling with the obturator nerve supply the obturator artery and the skin of the medial thigh. Sympathetic denervation of vessels in the lower limb can be effected by removing or ablating the upper three lumbar ganglia and intervening parts of the sympathetic trunk; this procedure may be useful in treating some varieties of vascular insufficiency of the lower limb. Pelvic sympathetic system the sacral region of the sympathetic trunk usually consists of four or five ganglia located medial or anterior to the anterior sacral foramina beneath the presacral fascia (Oh et al 2004). The sacral sympathetic trunk is continuous above with the lumbar sympathetic trunk, and preganglionic fibres descend from the lower lumbar spinal cord segments via this route. The sacral sympathetic chain is often asymmetric, with absent or fused ganglia, and crosscommunications between each side are frequent. Each ganglion sends at least one grey ramus communicans to its adjacent spinal nerve but up to 11 such branches from a single ganglion have been reported (Potts 1925). The pelvic sympathetic chain converges caudally to form a solitary retroperitoneal structure, the ganglion impar (or ganglion of Walther), which lies at a variable level between the sacrococcygeal joint and the tip of the coccyx; it is occasionally paired, unilateral or absent (Oh et al 2004). It conveys sympathetic efferents to and nociceptive afferents from the perineum and terminal urogenital regions. Ganglion impar blockade may be used to treat intractable perineal pain of sympathetic origin in patients with pelvic cancers (Toshniwal 2007). The postganglionic fibres are distributed via the sacral and coccygeal plexuses (Woon and Stringer 2014). Thus, sympathetic fibres in the tibial nerve are conveyed to the popliteal artery and its branches in the leg and foot, whilst those in the pudendal and superior and inferior gluteal nerves accompany these arteries to the perineum and buttocks. Pelvic splanchnic nerves Sacral splanchnic nerves Sacral splanchnic nerves pass directly from the ganglia to the inferior hypogastric plexus and, from there, to pelvic viscera; they usually arise from the first two sacral sympathetic ganglia. Parasympatheticinnervation the parasympathetic neurones innervating the abdomen and pelvis lie either in the dorsal motor nucleus of the vagus nerve or in the intermediolateral grey matter of the second, third and fourth sacral spinal segments. The vagus nerves supply parasympathetic innervation to the abdominal viscera as far as the distal transverse colon, i. The hindgut is supplied by parasympathetic fibres travelling via the pelvic splanchnic nerves (see below); the overlap between these two supplies is variable. The anterior vagal trunk is mostly derived from the left vagus and the posterior from the right vagus. The anterior trunk gives off a hepatic branch, which innervates the liver parenchyma and vasculature, the biliary tree including the gallbladder, and the structures in the free edge of the lesser omentum. The posterior trunk supplies branches to the coeliac plexus; these fibres frequently constitute the largest portion of the fibres contributing to the plexus. They leave the nerves as they exit the anterior sacral foramina and pass in the presacral tissue as a fine network of branches that are distributed to three principal destinations. Most pass anterolaterally into the network of nerves that form the inferior hypogastric plexus; from here, they pass to the pelvic viscera. Some join directly with the hypogastric nerves and ascend out of the pelvis, as far as the superior hypogastric plexus; from here, they are distributed with branches of the inferior mesenteric artery (p. A few run superolaterally in the presacral tissue, over the pelvic brim anterior to the left iliac vessels, and pass directly into the tissue of the retroperitoneum and the mesentery of the sigmoid and descending colon. The pelvic splanchnic nerves are motor to the smooth muscle of the hindgut and bladder wall, supply vasodilator fibres to the erectile tissue of the penis and clitoris, and are secretomotor to the hindgut. Coeliac plexus the coeliac plexus is located at the level of the twelfth thoracic and first lumbar vertebrae, and is the largest major autonomic plexus. It is a dense network that unites the coeliac ganglia, and surrounds the coeliac artery and the root of the superior mesenteric artery. Coeliac plexus Left greater splanchnic nerve Left lesser splanchnic nerve Right kidney Superior mesenteric plexus Left renal plexus Intermesenteric plexus Aorta and the beginning of the abdominal aorta, and lies medial to the suprarenal glands. The plexus and ganglia receive the greater and lesser splanchnic nerves and branches from the vagal trunks. Visceral afferents in the coeliac plexus convey pain and other sensations from upper abdominal viscera. Anaesthesia or ablation of these nerves (coeliac plexus block) is sometimes undertaken to treat intractable pain from pancreatic disorders. They vary in number, size, shape and precise location; there are often two, one on each side. The right ganglion is frequently posterior to the inferior vena cava, and the left ganglion often lies posterior to the origin of the splenic artery.
Cheap aceon 2 mg visa
Its numerous short fibres extend back from the posterior rough surface on the neck to the anterior surface of the transverse process arrhythmia management institute of south florida discount aceon 8mg without a prescription. A costotransverse ligament is rudimentary or absent in the eleventh and twelfth ribs. Superior costotransverse ligament the superior costotransverse ligament has anterior and posterior layers. The anterior layer is attached between the crest of the costal neck and lower aspect of the transverse process above, and blends laterally with the internal intercostal membrane; it is crossed by the intercostal vessels and nerve. The posterior layer is attached posteriorly on the costal neck, ascending posteromedially to the transverse process above, and blends laterally with the external intercostal muscle. The first sternocostal joint is an unusual variety of synarthrosis (fibrous) and is often inaccurately called a synchondrosis. The second to seventh costal cartilages articulate by synovial joints, although articular cavities are often absent, particularly in the lower joints. Fibrocartilage covers the articular surfaces and also unites the costal cartilages and the sternum in those joints where cavities are absent. Ligaments involved are capsular, radiate sternocostal, intra-articular and costoxiphoid. Contrast the concave facets on the upper transverse processes with the less curved facets on the lower transverse processes. Movements Costal heads are so firmly tied to vertebral bodies by radiate and intra-articular ligaments that only slight gliding can occur. The facets on the tubercles of the upper six ribs are oval and vertically convex, and fit corresponding concavities on the anterior surfaces of transverse processes; consequently, up and down movements of tubercles involve rotation of costal necks about their long axes. The facets on the seventh to tenth tubercles are almost flat and face down, medially and backwards; their opposing surfaces are on the upper aspects of transverse processes and so, when these tubercles ascend, they also move posteromedially. Both sets of joints move simultaneously and in the same directions; the costal neck therefore moves as if at a single joint in which the two articulations form its ends. In the upper six ribs, the neck moves slightly up and down but its chief movement is one of rotation about its long axis, which means that downward rotation of its anterior aspect is associated with depression, and upward rotation with elevation, of the shaft and anterior end of the rib. In the seventh to tenth ribs, the neck ascends posteromedially or descends anterolaterally, increasing or diminishing the infrasternal angle, respectively; slight rotation accompanies these movements. The lower border of transversus thoracis is in contact with the upper border of transversus abdominis in the interval between the sternal and costal origins of the diaphragm. They are thin, blended with the sternocostal ligaments, and strengthened above and below by fibres that connect the costal cartilages to the sternum. Costochondral junctions Artificially separated from its rib, a costal cartilage has a rounded end that fits a reciprocal depression in the rib. Periosteum and perichondrium are continuous across the costochondral junctions and the collagen of the osseous and cartilaginous matrices blend. Radiate sternocostal ligaments the radiate sternocostal ligaments are broad, thin bands that radiate from the anterior and posterior surfaces of the sternal ends of the costal cartilages of the true ribs to the corresponding sternal surfaces. Their superficial fibres intermingle with adjacent ligaments above and below, with those of the opposite side and with tendinous fibres of pectoralis major. Collectively, these tissues form a thick fibrous membrane around the sternum that is more marked inferiorly. Scapular muscles and muscles connecting the upper limb, chest wall and vertebrae, i. The ligament associated with the second costal cartilage extends from the costal cartilage to the fibrocartilage uniting the manubrium and sternal body, and is therefore intra-articular. Occasionally, the third sternal cartilage is connected with the first and second sternal segments by a similar ligament. Costoxiphoid ligaments Costoxiphoid ligaments connect the anterior and posterior surfaces of the seventh (and sometimes sixth) costal cartilage to the same surfaces of the xiphoid process. Movements Slight gliding movements, sufficient for ventilation, occur at sternocostal joints. Each articulation is enclosed in a thin fibrous capsule, lined by synovial membrane with lateral and medial interchondral ligaments. Sometimes the fifth cartilage, and less commonly the ninth cartilage, articulate at their inferior borders with adjoining cartilages; this connection is usually effected by ligamentous fibres. Articulation between the ninth and tenth cartilages is never synovial and sometimes absent. Each muscle passes from the lower border of one rib to the upper border of the rib below; their fibres are directed obliquely downwards and laterally at the back of the thorax, and downwards, forwards and medially at the front. In the upper two or three spaces, they do not quite reach the ends of the rib, and in the lower two spaces, they extend to the free ends of the costal cartilages. Muscles Innervation External intercostals are supplied by the adjacent intercostal nerves. Levatores costarum Action External intercostals are believed to act with the internal intercostals (Ch. They continue back as far as the posterior costal angles, where each is replaced by an aponeurotic layer, the internal intercostal membrane, that is continuous posteriorly with the anterior fibres of a superior costotransverse ligament, and anteriorly with the fascia between the internal and external intercostal muscles. Each muscle descends from the floor of a costal groove and adjacent costal cartilage, and inserts into the upper border of the rib below; their fibres are directed obliquely, nearly at right angles to those of the external intercostal muscles. Levatores costarum are strong bundles, 12 on each side, which arise from the tips of the transverse processes of the seventh cervical and first to eleventh thoracic vertebrae. They pass obliquely downwards and laterally, parallel with the posterior borders of the external intercostals. Each is attached to the upper edge and external surface of the rib immediately below the vertebra from which it takes origin, between the tubercle and the angle (levatores costarum breves). Each of the four lower muscles divides into two fasciculi; one is attached as already described, and the other descends to the second rib below its origin (levatores costarum longi). Innervation Levatores costarum are supplied by the lateral branches of the dorsal rami of the corresponding thoracic spinal nerves. Action Levatores costarum elevate the ribs but their importance in ventilation is disputed. They are also said to act from their costal attachments as rotators and lateral flexors of the vertebral column. Innervation Internal intercostals are supplied by the adjacent intercostal nerves. Innermost intercostals the innermost intercostals were once regarded as internal laminae of the internal intercostal muscles, and fibres in the two layers do coincide in direction. They are insignificant, and sometimes absent, at the highest thoracic levels but become progressively more substantial below this, typically extending through the middle two quarters of the lower intercostal spaces. Posteriorly, the innermost intercostals, in those spaces where they are well developed, may come together with the corresponding subcostales. The innermost intercostals are related internally to the endothoracic fascia and parietal pleura, and externally to the intercostal nerves and vessels. It arises by a thin aponeurosis from the lower part of the nuchal ligament, the spines of the seventh cervical and upper two or three thoracic vertebrae, and their supraspinous ligaments. It descends laterally and ends in four digitations attached to the upper borders and external surfaces of the second, third, fourth and fifth ribs, just lateral to their angles. It is superficial to the thoracic part of the thoracolumbar fascia and deep to the rhomboids. The number of digitations can vary from three to six, and the muscle may even be absent. Innervation Serratus posterior superior is innervated by the second, third, fourth and fifth intercostal nerves. Action the attachments of serratus posterior superior clearly indicate that it could elevate the ribs; its role in humans is uncertain. Innervation Innermost intercostals are supplied by the adjacent intercostal nerves. Semispinalis capitis Mastoid process Splenius capitis Action Innermost intercostals are believed to act with the internal intercostals (Ch. Subcostales Innervation Subcostales are supplied by the adjacent intercostal nerves. It arises from the lower third of the posterior surface of the sternum, the xiphoid process and the costal cartilages of the lower three or four true ribs near their sternal ends. The fibres diverge and ascend laterally as slips that pass into the lower borders and inner surfaces of the costal cartilages of the second, third, fourth, fifth and sixth ribs.
Purchase aceon with paypal
The fetal lung is a net fluid secretor heart attack 2o13 buy aceon visa, the output of fluid reaching as high as 5 ml/kg shortly before birth (see p. Most of the fluid produced within the lungs remains there because of the mechanical effect exerted by amniotic fluid pressure and, normally, only a small amount of this fluid contributes to the amniotic fluid. The normal functioning of the kidneys regulates the volume and pressure of the lung airway fluid and may, in turn, provide the pressure needed for expansion and enlargement of the bronchial and pulmonary systems. Interestingly, obstruction to the fetal airway causes accelerated maturation of alveoli; this approach has been used therapeutically in congenital diaphragmatic hernia, where intermittent inflation and deflation of an intratracheal balloon in a lamb model resulted in better lung growth and maturation (Nelson et al 2005). Two other cell types that are part of normal lung development, neuroendocrine cells and glycogen-containing cells, are implicated in postnatal paediatric interstitial lung disease, with early onset of respiratory distress. The earliest major effect is congenital diaphragmatic hernia, which leads to both ipsilateral and contralateral anomalies in branching pattern in the first 16 weeks of gestation that cannot be corrected subsequently, even by intrauterine tracheal balloon occlusion. Inevitably, abnormal branching leads to abnormal alveolarization in this condition. The most important later influence is maternal smoking but there is evidence that maternal exposure to pollution is also important (Latzin et al 2009). Animal studies have confirmed that antenatal nicotine exposure leads to structural changes in the fetal lung (Elliot et al 2001, Sekhon et al 2002, Fu et al 2011). Other factors that may be important include maternal hypertension in pregnancy, and maternal antibiotic and paracetamol usage. Ultrasound antenatal imaging of the lungs Pulmonary hypoplasia, an absolute decrease in lung volume and weight for gestational age, may be identified but cannot be confirmed. Unilateral hypoplasia may be suggested by rotation and displacement of the heart on ultrasound examination. In bilateral pulmonary hypoplasia, the heart appears relatively large with reduced chest volume. Increased echogenicity of the lungs indicates conditions that prevent the normal circulation of lung fluid (for further details, consult Coady and Bower (2015)). Details of prenatal lung anomalies and their detection are given in Epelman et al (2013). Virtually all the venous return to the right heart is shunted away from the lungs through the foramen ovale and the ductus arteriosus. At birth, the umbilical cord is tied and cut; within seconds, the lungs must take over all respiratory function in order to maintain life. However, it is clear that the first breath results in vasodilation by at least two mechanisms. First, there is the mechanical effect of traction on the vasculature: as the chest wall expands, it pulls open the pulmonary vessels. Second, oxygen entering the lungs for the first time produces pulmonary vasodilation. The gene for cyclo-oxygenase 1 (but not 2) in endothelium and vascular smooth muscle shows enhanced expression in late fetal and early postnatal life. Endothelin receptor expression increases around the time of birth, implying a role for this system in postnatal adaptation (Levy et al 2005). The role of nitric oxide in postnatal adaptation is controversial and there are likely to be important species differences. It is probable that the rapid deposition of type I collagen postnatally contributes structural stiffness to the blood vessel walls. Lung volume increases most rapidly during the first year of life and also increases more rapidly than airway calibre during this time, a finding consistent with the concept of dysanaptic lung growth, i. Until recently, it was thought that most of the alveoli were formed by 2 years of age, and that subsequent lung growth primarily resulted from an increase in the size of individual alveoli. However, recent work measuring alveolar size throughout childhood and adolescence using hyperpolarized helium has shown that neo-alveolarization, as well as increase in alveolar size, continues through childhood and adolescence (Narayanan et al 2012). This has important implications for diseases characterized by neonatal arrest of alveolarization. Forced vital capacity is reported to be the same in male and female infants and very young children (Jones et al 2000), which is consistent with postmortem morphometric measurements. By the second year of life, the rate of increase in forced vital capacity is similar to that reported in older children. During childhood, lung volumes and flow rates increase linearly with height, with a greater intercept and more positive slope in boys compared to girls (Stanojevic et al 2008). The linearity of these changes is interrupted by puberty (Rosenthal et al 1993a, 1993b, Rosenthal and Bush 2002), when important shape changes occur in the thorax, particularly in boys; these are mirrored by changes in the size of the underlying lung (and, in particular, the alveoli), as manifested by an abrupt increase in vital capacity, forced expired volume in 1 second, and total lung capacity. High-resolution computed tomographic scanning has been used to measure airway dimensions (de Jong et al 2006), although this technique should be used with caution because of the radiation exposure. At birth, the internal elastic lamina of the small muscular arteries consists only of amorphous elastin in a basal lamina-like matrix. By 3 weeks of age, a definitive elastic lamina is evident, although it is heavily fenestrated, permitting contact between the endothelial cells and the smooth muscle cells. The intrapulmonary arteries increase in size and their walls increase in thickness. However, the maturation of all of the pulmonary vascular smooth muscle cells, from the hilum to the precapillary bed, is not advanced until 2 years. As the distal airspaces expand, the capillary nets fuse from one alveolus to another, forming, for a period, an extensive double capillary net; this process can be seen from postnatal day 28, becomes more extensive by 1. Normal postnatal pulmonary arterial development Immediately after birth, dramatic remodelling of the pulmonary vasculature occurs, to effect an abrupt reduction of pulmonary vascular resistance. Failure to remodel in the presence of an anatomically normal heart leads to persistent pulmonary hypertension. Normal postnatal pulmonary arterial development in the full-term neonate can be divided into three stages. Adverse effects on postnatal lung development Stage one this lasts from birth to about postnatal day 4 and concerns the immediate adaptation to extrauterine life. At birth, the endothelial cells of the precapillary arteries are squat and have narrow bases on the subendothelium, a low surface to volume ratio and many surface projections. The smooth muscle cells show a significant reduction in diameter during this time. Current extremely premature babies, who are treated with surfactant and ventilated using low pressure, high-risk strategies, have arrest of alveolar development and pulmonary hypoplasia. Survivors from the pre-surfactant era have disease dominated by airflow obstruction. Postnatal lung development is also impaired by exposure to tobacco smoke, and there is increasing evidence that air pollution (Schultz et al 2012) and early sensitization to aeroallergens (Illi et al 2006) also have an adverse impact on normal lung growth. In addition, congenital anomalies of the heart and great vessels, and of the chest and abdominal walls, including neuromuscular disease, may impact on lung development. It is somewhat artificial to describe airway malformations in isolation, and the possibility of associated vascular anomalies must always be considered. Moreover, descriptions of what is seen clinically should be kept separate from speculations about the embryological origins of the anomaly. A review of all aspects of the diagnosis, nomenclature and management of congenital malformations of the airways, lung and chest wall. A review of how early problems with lung growth and development impact on later lung disease. Bush A 2009 Prenatal presentation and postnatal management of congenital thoracic malformations. An explanation of how tracheal occlusion can be used in an animal model to improve lung growth and maturation. Tracheobronchomalacia usually presents in early infancy with cough, tachypnoea, stridor and wheeze; it may also be associated with cardiac or respiratory anomalies, such as absent pulmonary valve syndrome, or tracheo-oesophageal fistula, respectively.


